Capillaroscopy is a method of recognized value in the study of morphological and functional abnormalities of the microcirculation. Its use in the clinical field dates from the early twentieth century,1 extending the clinical applications gradually over the years,2,3 to be considered a “routine investigation” in (nowadays) rheumatological practice.
The introduction of videocapillaroscopy with optical probes in contact resulted in a significant leap forward in the study of microcirculation.4,5
Videocapillaroscopy is indicated in all diseases whose pathogenesis recognizes anatomical and/or functional abnormalities in microcirculation.6
The advantages of capillaroscopy can be identified in the absence of invasiveness, in its remarkable sensitivity and specificity together with ease procedure and speed execution time. The relatively videocapillaroscopy low cost could be considered another big advantage of this procedure.
Videocapillaroscopy allows clinicians to acquire informations on morphological and hemodynamic parameters that could be useful like predictive value of extension, gravity and evolution of disease. In this sense videocapillaroscopy could be considered a double profile examination, with diagnostic and prognostic values in the same time.7
Among the many diseases related with abnormal capillaries there is diabetes mellitus. The interest of the microcirculation being altered glucose metabolism is well documented both in the course of diabetes type I and type II.
Chronic hyperglycemia is responsible for the metabolic alterations in endothelial cells, and also of the smooth muscle of the vessel walls, resulting in impairment of contractile properties together with permeability and hemodynamic modifications.
Parietal elasticity alteration transforms the vases in rigid ducts by thickened walls, partly due to the non-enzymatic glycosylation of proteins and partly due to the altered metabolism of myo-inositol.8
Decreased of parietal compliance cause the appearance of ectasia, with microaneurysms related to pressor stress.
In the context of diabetic angiopathy we can distingue nonspecific changes especially at the level of the arteries of large- and medium-caliber, and more specific, also if not exclusive, changes usually localized in the microvessel.
Microvessel videocapillaroscopy study seems to be very interesting in diabetes because microangiopathy is the most common and fearsome complication of diabetes and because it seems that the alterations of cutaneous microvessels and of the fundamental substance of connective constitute the earliest manifestations of the disease.
Modifications of cutaneous microvessel results in thickening of the capillary basement membrane, endothelial cells proliferation and vascular leakage.
Vascular leakage, detectable by fluorescence videocapillaroscopy, only in part seems to be related to an increase in the amplitude of the system to “small pores”, responsible for the trans-capillary diffusion. In fact, the accumulation of mucopolysaccharides, to which depends the thickening of the basal membrane, also change the selectivity of the process of filtration.9
The capillary flow appears grainy and slowed by the presence of microaggregates which often interrupt the continuity of the blood stream. This dynamic alteration is the consequence of an increased friction, in part connected to the marked tortuosity of the capillaries, in part to the increase of blood viscosity and platelet adhesiveness.10
The data so far in the literature about the capillaroscopic characteristics of diabetic microangiopathy are discordant. It ‘was reported a provision of the capillaries to “shoal of fish” in diabetes mellitus type I and increased morphological variability of the loops with increasing diameter.
A further and interesting capillaroscopic modification consists in a progressive dilation of efferent portion of the loop, homogeneous and centripetal, such as to give the appearance to capillary classic “elephant nose,” (Figures 1 and 2) provided that the diameter exceeds at least 5 times the physiological value (7-18 microns according Grassi) and that it is repetitive.
Figure 1: Elephant nose.
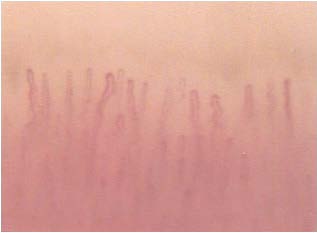
Figure 2: Elephant nose.
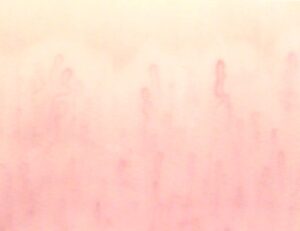
Other capillaroscopic alterations consist of sacculare aneurysm apical and lateral of the loops. (Figures 3 and 4) Arborescent aspects, expression of neo-vascularization, and microhaemorrhages more or less pronounced, associated with slowing and grain flow, (Figures 5-7) are interpreted as an expression of greater severity of microangiopathy.11
Figure 3: Sacculare aneurysm apical.
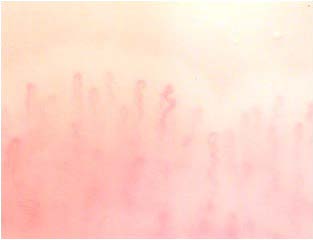
Figure 4: Sacculare aneurysm lateral.
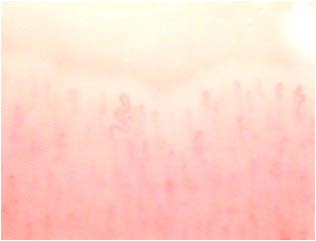
Figure 5: Arborescent aspects
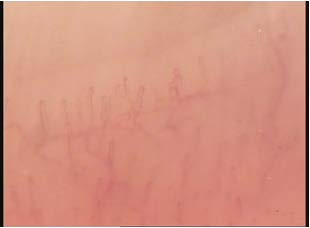
Figure 6: Microhaemorrhages.
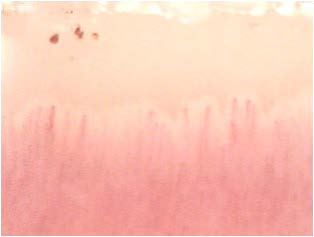
Figure 7: Microhaemorrhages.
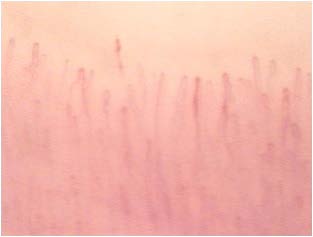
Although not yet encoded a capillaroscopic pattern characteristic, in patients with diabetes mellitus can detect morphological expression of microangiopathy such as, for example, the homogeneous increase of the diameter of the capillaries (especially at the level of the stretch venular) and the presence of loops of look convoluted. (Figures 8-11).
Figure 8: Diameter exceeds.
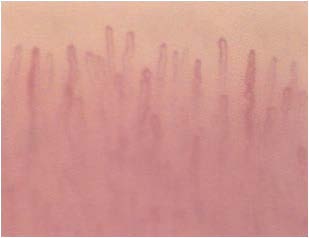
Figure 9: Diameter exceeds.
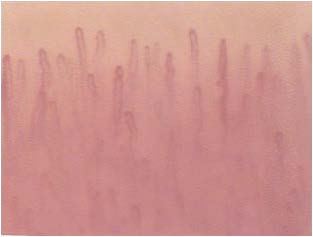
Figure 10: Loops convoluted.
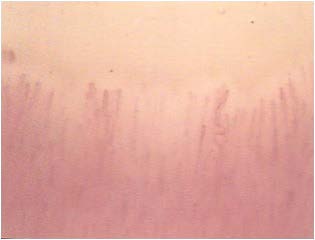
Figure 11: Loops convoluted.
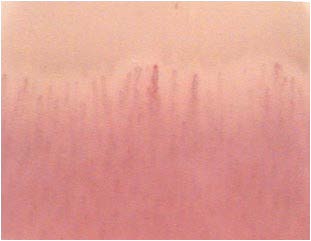
These anomalies, although not of specific diagnostic value, can play a significant role in the characterization of diabetic microangiopathy.12
With the use of fluorescenze capillaroscopy was detected a difference between the permeability alterations typical of diabetes mellitus and those features of systemic sclerosis.13 In the first case, in fact, the alteration of the permeability is homogeneously distributed throughout the course of the loop capillary (venular and arteriolar tract).
In scleroderma, however, the defect is asymmetric and is accompanied by loss of focal tracer.14 The analogy between two seemingly distant disease, such as diabetes and systemic sclerosis, supports the hypothesis already advanced from the Jordan (“diabetes as a disease of the connective deposit”) of a final common pathway of damage microcirculatory unit, regardless of the initial pathogenic noxa.15
CONFLICTS OF INTEREST
None.
















