INTRODUCTION
Ventricular septal rupture (VSR) is a rare and life-threatening complication of acute myocardial infarction (AMI). The advanced age, female sex, stroke history, chronic kidney disease (CKD) and heart failure are the independent risk factors for rupture in patients presenting with AMI.1 VSR is usually concomitant with the conditions such as ST-segment elevation, initially positive cardiac biomarkers, cardiogenic shock, high Killip class, excessive time spent till ballooning or thrombolytic application.1 Rupture develops in any anatomical locations of the ventricular septum and at similar frequencies after anterior, inferior and lateral transmural infarctions.1 Anterior infarction tends to lead apical defects, whereas inferior or lateral infarctions lead more frequently to defects in posterior wall or basal septum.1
CASE REPORT
A 66-year-old male patient complaining of epigastric pain starting the night before was brought to the emergency room by his relatives. The patient’s medical and family history did not feature anything but hypertension for ten years. His vital signs were as follows: pulse rhythmically 120/min, arterial blood pressure 100/60 mmHg, respiratory rate 30/min, and body temperature 36.5 °C. The patient being good in general condition revealed rales in the basal lungs in pulmonary auscultation, and 2/6 systolic murmur radiating to left axilla in cardiac auscultation. There was no pathology in the other system examinations of the patient. The electrocardiogram was 120/min in rate with sinus rhythm and ST-segment elevation was detected in the lead II, III, and aVF, and ST-segment depression in the lead V1-V3, also revealing reciprocal ST-segment depression in the lead I-aVL, V4-V6 suggestive of inferoposterior myocardial infarction (Figure 1).
Figure 1: Detection of Ventricular Septal Supture by Echocardiography.
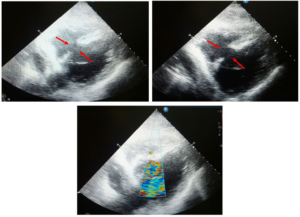
The echocardiography revealed EF 45% and VSR in the basal septum. The patient who received initial treatment for AMI was referred to an advanced center for coronary angiography. The patient was admitted as exitus on the post-operative second day of admission to the intensive care unit (ICU).
DISCUSSION
AMI and infarction-related complications are the major causes of death today.2 Post-infarction VSR is usually seen after the first AMI in ones without known coronary artery disease.2 The first finding in cases with post-infarction rupture is usually the pansystolic murmur heard in mesocardiac area and left sternal border between the 2nd and 4th days of the infarction.2 There are no specific electrocardiogram (ECG) features for post-infarction VSR. However, some studies demonstrated that persisting ST-segment elevation >72 hours, right bundle branch block (RBBB), left bundle branch block (LBBB), left anterior fascicular block (LAFB), left posterior fascicular block (LPFB), advanced atrioventricular blocks, ventricular and supraventricular tachycardias, and RV extension may be suggestive of post-infarction VSR.3 In our case, there was no specific ECG finding suggestive of post-infarction VSR (Figure 1). VSR has become a rare complication thanks to the acute reperfusion strategies applied after myocardial infarction.1 While its incidence was 1-2% in the pre-thrombolytic period, this rate was reduced to 0.2% by using thrombolytic agents.4 In a study by Harlan et al5 in 2006, the 30-day mortality rate for AMI in the United States was 16.1%, and in-hospital mortality rate was 10.1%. Once post-infarction VSR ensues, mortality is quite high despite the improvement in surgical technique, invasive cardiology, and drug therapy. While the mortality rate is 90-95% by conservative treatment, it ranges between 19-60% by surgical approach.4 Mortality rate of VSR vary among the centers.4 Inferior myocardial infarction, cardiogenic shock, right ventricular dysfunction, two or three-vessel disease, not to take thrombolytic therapy, and length of time to surgery are the main determinant factors for prognosis in ones with rupture.4 Operative intervention is most successful in patients with STEMI and circulatory collapse when a surgically correctable mechanical lesion such as ventricular septal defect or mitral regurgitation can be identified and repaired. In such patients, the circulation should at first be supported by intra-aortic balloon pulsation and a positive inotropic agent such as dopamine or dobutamine in combination with a vasodilator, unless the patient is hypotensive. Surgery should not be delayed in patients with a correctable lesion who agree to an aggressive management strategy and require pharmacologic and/or mechanical (counterpulsation) support. Such patients frequently develop a serious complication (e.g., infection, adult respiratory distress syndrome, extension of the infarct, renal failure) if surgery is delayed. Surgical survival is predicted by early operation, short duration of shock, and mild degrees of right and left ventricular impairment.6,7 When the hemodynamic status of a patient with one of these mechanical lesions complicating STEMI remains stable after the patient has been weaned from pharmacologic and/or mechanical support, it may be possible to postpone the operation for 2 to 4 weeks to allow some healing of the infarct to occur.
Transcatheter closure of post-infarction VSR as a less invasive approach may improve survival rates in selected patients with suitable anatomy.8 Immediate reduction of the left to-right shunt, even if the VSR is not completely closed, may stabilize the patient enough to function to bridge to surgery.9 In patients undergoing cardiac surgery, pre-operative antiplatelet administration increases post-operative bleeding and red blood cell requirements with no effect on mortality, re-exploration rate, and peri-operative MI.10 A new meta-analysis concluded that antiplatelet therapy was associated with increased chest tube drainage in this patient population and might be associated with a greater requirement for blood products.11 However, in patients undergoing transcatheter VSR repair, dual antiplatelet therapy and heparin may be used at identical risks as the patients undergoing procedures with and without PCI.
Medical management is not a feasible therapeutic option for most patients; however, it is aimed at decreasing afterload with medications or support devices and therefore left-to-right shunting.1 Hence, especially the patients with unstable hemodynamics should immediately be transferred to an advanced center with capability of cardiac surgery.
As a result, special attention should be paid to complications in patients admitted to the emergency room with myocardial infarction.
CONFLICTS OF INTEREST
The authors declare that they have no conflicts of interest.
CONSENT
The authors have received written informed consent from the patient.






