INTRODUCTION
The main goal of diabetes treatment is to maintain an adequate metabolic control in order to prevent or delay disease complications.1 However, when too strong efforts are made to keep glycated haemoglobin (HbA1c) within target levels, patients often are at a high risk for hypoglycaemia.2 Hypoglycaemic episodes (Hypos), especially severe ones, have a relevant clinical, social and economic impact. From a clinical standpoint, they are characterized by several symptoms ranging from general discomfort to seizures and coma eventually resulting into either sudden cardiac arrhythmic death or lethal brain damage due to long-lasting cerebral tissue glucose deprivation.3 Repeated severe Hypos are associated with a higher cardiovascular risk,4 and dementia.5,6 Even mild symptomatic hypoglycaemia is associated with an increased risk of cardiovascular events and all-cause hospitalization/mortality.7
Hypoglycaemia is also a key driver for direct and indirect costs. In greater detail, up to one third of people with long standing type 1 diabetes mellitus (T1DM) and one fifth of insulin treated people with type 2 diabetes mellitus (T2DM) suffer from one or more severe Hypos every year.8,9 This generates a high economic burden in terms of both direct (>3000 € each hospital admission all over Europe) and indirect costs (mostly due to patients or their relatives abstaining from work).10,11
Moreover, from a social point of view, several studies suggest that Hypos may disrupt quality of life (QoL) in terms of driving efficiency, work performance and recreational pursuits 12 and thereby negatively affect patient’s mood and health perception.13,14 In addition, Hypos are by far the strongest factors hindering metabolic control in insulin-treated patients 15,16 as long-lasting hyperglycaemia invariably follows Hypos due both to rebound counter-regulatory hormone reaction and to previous frightening experiences causing excessive corrective behaviour and poor adherence to therapy. As a consequence of that high glucose variability (GV) is also often observed.17,18 Iatrogenic causes explain nearly all Hypos in people with T1DM.19,20 Predictable triggers (i.e. skipped meals, strenuous exercise, insulin overdose, neoplasms and chronic renal or liver disease), as well as, defective glucose counter-regulation and established hypoglycaemia-associated autonomic failure have been extensively described.21,22,23
On the other hand, many Hypos are difficult to explain on the basis of the above listed factors and might also depend on improper insulin administration. This includes either accidental intramuscular injection through excessively long needles or bad patients’ habits, like shooting insulin into skin nodules.24,25,26 Diabetologists spend too little time in educating patients to perform correct insulin injections27 and, as a consequence of that, several groups have being describing a close relationship between recurrent hypoglycaemia and lipohypertrophy (LH) so far.27,28,29,30 In fact, patients and clinicians are not used to systematically and thoroughly investigate upon any possible reasons behind unexplained Hypos.
The main purpose of this study was to assess whether LH occurrence (Figure 1) might predict a high Hypos or GV rate in insulin treated patients and, if so, whether high Hypo rates might depend on inappropriate injection habits like performing insulin shots into LH nodules with long needles.
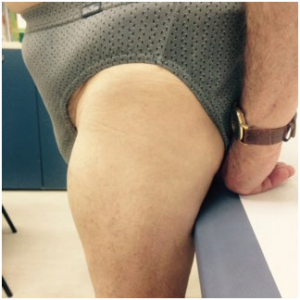
Figure 1: The red arrow points to a prominent LH nodule on the left thigh of a woman with T1DM.
PATIENTS AND METHODS
This was an observational, retrospective, multicentre study on T1 or T2DM patients referring to five diabetes outpatient centres.
Eligibility criteria were: age >18 years, insulin treatment (>2 daily shots) for at least the last 12 months, as well as, frequent unexplained Hypos and/or unjustified GV over the last three months.
Exclusion criteria: Hospitalization, use of insulin syringes, oral hypoglycaemic agents or steroids, neoplastic, liver or kidney diseases, pregnancy.
The study was approved by the Ethics Committee of the five participating centres and all patients agreed to participate by signing their informed consent to the study. The latter was performed according to the Helsinki Declaration.
All enrolled patients filled in the HYPOS-1 questionnaire to collect information on Hypos or GV as previously described.31,32 Briefly, the questionnaire investigated upon sociodemographic and clinical characteristics including age, gender, education level, employment, living status, spouse/caregiver support, number of daily medications (other than blood glucose lowering agents), number of severe hypoglycaemic episodes during the past 12 months, and number of symptomatic Hypos in the past 4 weeks. The above mentioned time intervals were chosen according to the ability of insulin treated subjects to recall such episodes as ascertained in previous studies.23 Study investigators completed a web-based electronic clinical record form (eCRF) where patients were identified by a unique ID number, which was also used as a key linkage to merge their data with questionnaire answers and to anonymously analyse them thereafter.
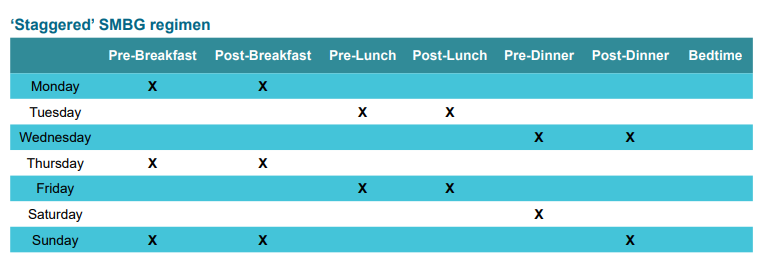
The evaluation-validation of Hypos and GV was based on the analysis of patients’ self-monitored blood glucose (SMBG) recordings using a twice-a-day “staggered’’ scheme as depicted in Figure 2. This pattern is characterized by high flexibility, which makes it better tolerated and accepted with consequent greater patient adherence to given instructions, and is strongly supported by specialists, as well, because of its easier and immediate interpretation.24 Patients were also trained to perform additional glucose checks in case of sudden hypoglycaemic symptoms.
Figure 2: Self-monitoring blood glucose tabs weekly grid, according to the IDF 2015 scheme.
In greater detail:
1. Hypo was defined as the occurrence of one or more symptoms of hypoglycaemia (such as palpitations, tiredness, sweating, hunger, dizziness and tremor) and a confirmed blood glucose (BG) meter reading of ≤70 mg/dL, according to ADA statements.15 Frequent unexplained hypoglycaemia was defined as having a Hypo atleast once a week in the absence of any identified precipitating event, such as changes in insulin dosage, diet composition or amount of physical activity. Hypos were further defined as severe (SH; BGB<50 mg/dl), and non-severe (NSH; 50th>BG<70 mg/dl).33,34,35
2. GV was investigated by a validated questionnaire already used in previous studies.28,31,36 As GV has no universally accepted definition, patients were defined as having GV if blood glucose values unpredictably and inexplicably swung from 250 mg/dL on a continuous basis at least three times a week.28,31,36
3. LH was looked for at all insulin injection sites using a previously described validated methodology: when filling in their questionnaires patients also indicated all sites they used35 and the medical staff checked them for the presence of skin lesions according to a structured protocol, as previously described.37,38
Participating centres screened 3710 consecutive ≥18 year old outpatients treated with insulin pens for at least two years and free from any kind of (i) steroid-based treatment regimens, (ii) chronic kidney/liver disease or (iii) cancer. Two thousand three hundred out of them (62%) had experienced at least one episode of either SH or NSH over the last three months. After excluding all cases associated with factors known to cause hypoglycaemia, including dietary errors, inadequate insulin dosage, poor treatment adherence, occasional utilization of drugs endowed with intrinsic hypoglycaemic effects, acute gastrointestinal troubles, only 387 subjects (16.8%) with unexplained GV and/or Hypos were finally included in the study: 294 had both Hypos and GV, 23 only Hypos and 70 only GV. In line with the typical distribution among people with diabetes attending such centre, 80 out of the 387 enrolled subjects had T1DM. Main clinical participant characteristics are described in Table 1, whereas needle length and gauge are summarized in Table 2. Participants using premixed or NPH insulin (9.3% and 2.8%, respectively) or 12 mm length and 29 gauge needles (1.8% each) turned out to be only a minority and were therefore excluded from subsequent statistical evaluation to avoid any bias due to treatment in homogeneity. No patient used any 33 gauge needles.
Table 1: Characteristics of insulin pen using patients.
|
Subjects enrolled
|
n=387 |
| Male gender (%) |
45.5
|
|
Age (years) (range)
|
61±16 41-77 |
| BMI (Kg/m2) |
29±8
|
|
Type 1 diabetes n.(%)
|
81 (20.9%) |
| HbA1c (%) |
8.4±1.2
|
|
DM duration (years) (range)
|
13±9 5-23 |
| Injections/day (n.) |
3.8±1.2
|
|
Insulin treatment (years)
|
10±9 |
| Insulin dosage (IU/day) |
44±25
|
|
Basal insulin analogues n.(%)
|
340 (87.9%) |
| Short-acting insulin analogues n.(%) |
360 (93.0%)
|
|
Premixed Insulin n.(%)
|
36 (93%) |
| Insulin–NPH n.(%) |
11 (2.8%)
|
Statistical Analysis
Continuous variables were reported as means±standard deviation (SDs) and were compared between groups by student’s t-test for independent samples or analysis of variance (ANOVA) as needed. Non-parametric Mann-Whitney U-test was used when appropriate. Categorical variables were summarized as rate or percentage and their bivariate associations were evaluated by Chi-square or Fisher exact tests. Based on the fact that a significant association had been already documented in the Hypos-1 study33,34 among the presence of SH/NSH and diabetes complications, type of insulin preparation or treatment with drugs associated with reduced Hypo awareness including angiotensin-converting-enzyme inhibitor (ACE inhibitor) or beta-blockers,39,40,41 we also evaluated the relationship between LH lesions and these factors based on univariate followed by Poisson multivariate model analysis. Patients were grouped and compared according to their LH identification rate characteristics, namely by considering those identified by all health providers (HPs) vs. those identified by none of them. Odds ratios (ORs) for LH were reported along with their 95% confidence intervals (CI). In addition, the relationship between needle length/gauge and the presence of LH was also evaluated according to the same statistical procedure by taking into account school education, employment and marital status or living conditions. The analysis were carried out using STATA software, version 12 (Stata Corp, College Station, TX, USA) and p-values<0.05 were accepted a priori as statistically significant.
RESULTS
General patient characteristics are described in Table 1. Twentyeight percentages subjects had faced at least one SH, and 72% one NSH in the last four weeks, with an average of 3.3 episodes per week. Table 2 describes needle utilization rate as referred to length and gauge and shows that lengths >4-5 mm and gauges. <3were largely preferred. Table 3 clearly describes the relationship between unexplained GV/Hypos and LH (p<0.0001) characterized by a significantly high OR (7.18) for GV/Hypos in case of LH.
Table 2: Needle length and gauge.
|
length (mm)
|
Subjects n. (%) |
Gauge (G) |
Subjects n. (%)
|
|
4
|
58 (14.9) |
33 |
–
|
|
5
|
61 (15.8) |
32 |
58 (14.5) |
| 6 |
115 (29.7) |
31 |
176 (45.5)
|
|
8
|
146 (37.7) |
30 |
146 (37.7) |
| 12 |
7 ( 1.8) |
29 |
7 (1.8)
|
Table 3: Association between unexplained glycaemic variability (GV)/Hypoglycaemic episodes (Hypos) and lipohypertrophy (LH); n and (%) are displayed within cells (χ2 test, p<0.0001).
|
|
GV/Hypos
|
|
| LH |
|
yes |
no |
total
|
|
yes
|
287 (74.2%) |
11 (2.8%) |
298 |
| no |
7 (1.8%) |
82 (21.2%) |
89
|
|
total
|
294 |
94 |
387
|
Tables 4 and 5 describe the results of univariate analysis by comparing subjects with LH to those without LH regardless of general/demographic parameters, insulin treatment features, injection site rotation habits, as well as, needle reuse rate, utilization of drugs associated with reduced hypoglycaemia awareness, employment, school education, living conditions and marital status. In greater detail, 40% patients used the same needle at least twice and 44.2% at least three times; only 15.8% subjects changed needles at each injection. Tables clearly show that needle reuse and failure to rotate injection sites associated to a high LH risk.
Table 4: Univariate analysis of clinical parameters with respect to lipohypertrophy in insulin pen using patients.
From 387 subjects with hypoglycaemia and/or glycaemic variability, 23 were Hypos- and 70 were GV.
Because of the low number of subjects using premix or NPH insulin, these were not included in the statistical evaluation
| |
Lipohypertrophy
|
| Overall n=387 |
No n=89 (22.9%) |
Yes n=298 (77.1%) |
OR (95% CI) |
p
|
|
Male gender
|
187 (48.4%) |
87 (46.8%) |
100 (50.1%) |
1.20 (0.88-1.64) |
0.243 |
| Age (years) |
61±16 |
63±12 |
61±10 |
0.90 (0.81-1.00) |
0.051
|
|
BMI (Kg/m2)
|
29±6 |
29±6 |
29±6 |
0.90 (0.81-1.00) |
0.492 |
| Type 1 diabetes |
81 (20.9%) |
20 (25.2%) |
61 (75.3%) |
2.64 (2.14-2.74) |
<0.001
|
|
HbA1c (%)
|
7.8±1.3 |
7.5±1.1 |
8.3±1.2 |
1.30 (1.14-1.48) |
<0.001 |
| DM duration (years) |
17±10 |
17±9 |
20±8 |
1.28 (1.10-1.49) |
<0.001
|
|
Injections/day n.
|
3.8±2.8 |
3.7±3.5 |
3.7±.9 |
1.00 (0.94-1.06) |
0.991 |
| Insulin treatment (years) |
8±9 |
7±9 |
12±6 |
1.46 (1.23-1.74) |
<0.001
|
|
Insulin dosage (IU/day)
|
44±25 |
41±19 |
49±18 |
1.13 (1.06-1.21) |
<0.001 |
| Basal insulin analogues n. (%) |
340 (87.9%) |
238 (70.0%) |
102 (30.0%) |
3.13 (1.80-5.47) |
<0.001
|
|
Short-acting insulin analogues n. (%)
|
360 (93.0%) |
298 (82.8%) |
62 (17.2%) |
3.27 (2.68-3.78) |
<0.001 |
| Short-acting insulin analogues n. (%) |
364 (94.1)
|
109 (29,9) |
255 (70.1) |
4.43 (3.11-6.33) |
<0.001
|
| Hypoglycaemia n. (%) |
317 (81.9) |
27 (8.5) |
290 (91.5) |
5.23 (3.54-7.74) |
<0.001
|
|
Needle reuse n. (%)
|
326 (84.2) |
61 (18.7) |
326 (81.3) |
4.27 (3.89-4.81) |
<0.001 |
| No injection site rotation n. (%) |
349 (90.2) |
38 (10.9) |
349 (89.1) |
5.11 (4.76-5.56) |
<0.001
|
|
Drugs associated with reduced awareness (%)
|
|
ACE-Inhibitors
|
30 |
48 |
52 |
1.29 (0.82-1.74) |
0.219 |
| Beta-blockers |
25 |
47 |
51 |
0.98 (0.79-1.24) |
0.317
|
Table 5: Univariate analysis of needle length and gauge with respect to lipohypertrophy in insulin pen using patients.
|
Lipohypertrophy
|
|
Overall n=387
|
No n=89 (22.9%) |
Yes n=298 (77.1%) |
OR (95% CI) |
p
|
|
needle length n. (%)
|
| 4 mm |
58 (14.9%) |
47 (81.1) |
11 (18.9)
|
2.18 (2.02-2.56) |
<0.01 |
|
5 mm
|
61 (15.8%) |
50 (82.0) |
11 (18.0) |
2.27 (1.79-2.84) |
<0.01 |
| 6 mm |
115 (29.7%) |
44 (38.3) |
71 (61.7)
|
1.94 (1.11-2.73) |
<0.01 |
|
8 mm
|
146 (37.7%) |
31 (21.2) |
115 (78.8) |
2.22 (2.08-3.29) |
<0.01 |
| 12 mm |
7 (1.8%) |
0 (0.0) |
7 (100.0)
|
– |
– |
|
needle gauge n. (%)
|
|
32
|
58 (14,5) |
47 (81.1) |
11 (18.9) |
2.15 (1.95-2.48) |
<0.01 |
| 31 |
176 (45,5) |
85 (48.3) |
91 (51.7)
|
1.22 (0.89-1.67) |
0.257 |
|
30
|
146 (37,7) |
37 (25.3) |
109 (74.7) |
1.56 (1.20-1.78) |
<0.01 |
| 29 |
7 (1,8) |
0 (0.0) |
7 (100.0)
|
– |
– |
|
needle reuse n. (%)
|
326 (84.2) |
61 (18.7) |
326 (81.3) |
4.27 (3.89-4.81) |
<0.01 |
| no injection site rotation n. (%) |
349 (90.2) |
38 (10.9) |
349 (89.1)
|
5.11 (4.76-5.56) |
<0.01 |
T1DM, long standing disease and high insulin requirements significantly associated to LH, and very high ORs were attained by unexplained Hypos (4.43), GV (5.23) or both (7.18). The risk of diabetes complications was significantly higher in subjects with LH (retinopathy 4.12; nephropathy 4.19; lower limb complications 4.48; sensory-motor neuropathy 5.18, and autonomic neuropathy 3.72) (Figures 3 and 4). Any use of drugs associated with reduced hypoglycaemia awareness turned out to be irrelevant.
Figure 3: Frequency of LH and diabetes complications. All differences were statistically significant (p<0.001).
CV: Cardio-CerebroVascular complications; R: Retinopathy, Neph: Nephropathy; PA: Peripheral Artery disease; Per Neuro: Peripheral Neuropathy; Aut Neuro: Autonomic Neuropathy.
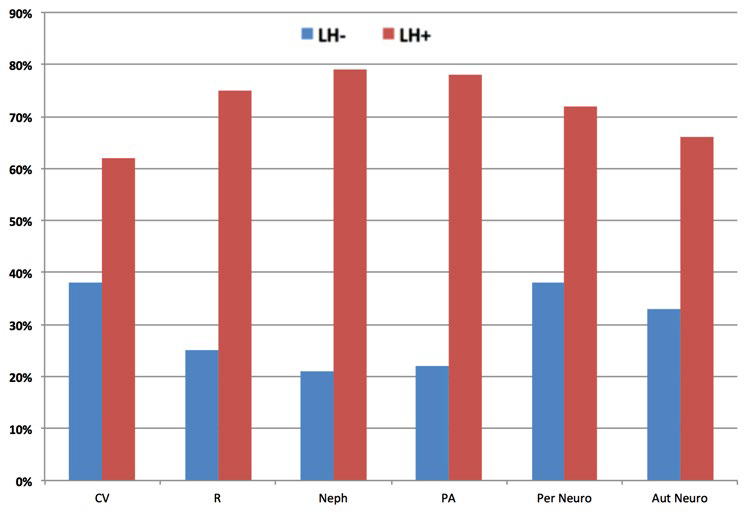
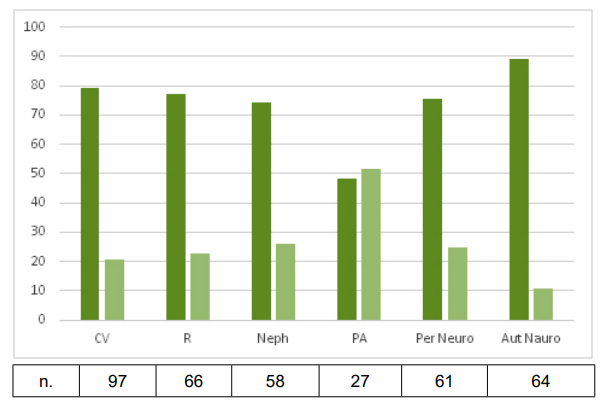
Figure 4: Complication rates in subjects with (dark green bars) or without (light green bars) hypoglycaemia/glycaemic variability; 57% had more than one complication and 32% more than two complications. All the differences were statistically significant (p<0.001) except for PA.
CV: Cardio-CerebroVascular complications; R: Retinopathy; Neph: Nephropathy; PA: Peripheral Artery disease; Per Neuro: Peripheral Neuropathy; Aut Neuro: Autonomic Neuropathy.
With reference to the socio-economic status, the level of education did not affect the association between Hypos and LH. The same applied to living conditions: in fact no significant association was found between Hypos and LH in those who lived alone, or were unmarried/separated/divorced.
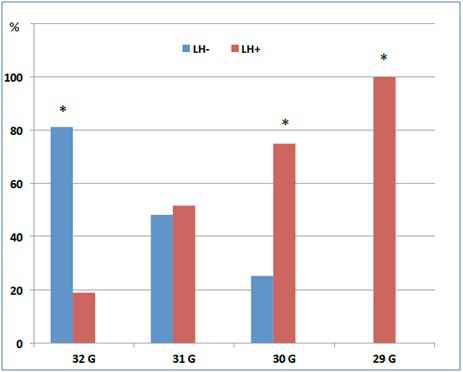
Finally, as shown in Figures 5 and 6, the risk of developing LH was associated with needle characteristics: both longer and larger needles were related to a higher LH risk (p<0.001). Needle reuse and missed injection site rotation were also LH risk factors strongly associated with unexplained Hypos /GV.
Figure 5: Relationship between needle length and lipohypertrophy. *p<0.001
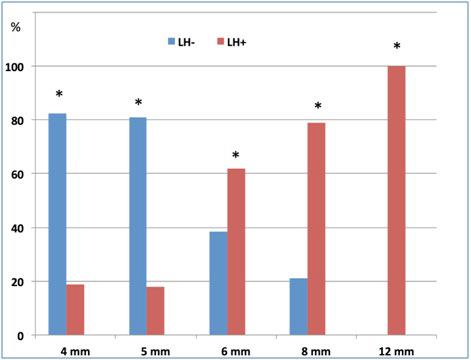
Figure 6: Relationship between needle gauge and lipohypertrophy. *p<0.001.
After multivariate analysis (Table 6) short-acting and basal insulin analogues kept highly associated with increased LH risk in T1DM patients with unjustified Hypos /GV.
Table 6: Multivariate analysis of subjects’ characteristics potentially associated to lipohypertrophy.
|
OR (95% CI)
|
p |
| Age (years) |
0.91 (0.76-1.10) |
0.338
|
|
Type 1 diabetes n. (%)
|
0.84 (0.39-1.80) |
0.656 |
| HbA1c (%) |
2.00 (1.85-2.19) |
<0.001
|
|
DM duration (years)
|
1.07 (0.80-1.44) |
0.627 |
| Insulin dose (IU/day) |
2.88 (2.56-2.99) |
<0.001
|
|
Hypos/GV
|
4.85 (4.02-5.30) |
<0.001 |
| Diabetes complications |
2.99 (2.34-3.51) |
<0.001
|
|
Living status
|
3.15 (2.89-3.58) |
<0.001 |
| Marital status |
3.22 (2.97-3.69) |
<0.001
|
|
Pen needle length (mm)
|
2.98 (2.56-2.25) |
<0.001 |
| Gauge (G) |
3.56 (2.60-3.78) |
<0.001
|
|
Needle reuse
|
3.78 (3.06-4.12) |
<0.001 |
| Failure to rotate injection sites |
4.22 (3.78-4.86) |
<0.001
|
When pooling socio-economic and family statuses under the term of living conditions and putting together all diabetes complications, we could also confirm both complex parameters to be strongly associated to LH and GV/Hypos. Likewise, needle length/gauge, needle reuse and missed injection site rotation kept their significant role as risk factors for LH in the presence of unexplained GV/Hypos. This was not the case, instead, with either disease duration or daily insulin dosage.
DISCUSSION
Hypoglycaemia is an acute complication increasing diabetes morbidity and mortality. It is also responsible for a heavy—still not fully defined-economic burden. However it is by far the strongest limiting factor in terms of tight glycaemic control in insulin-treated patients.17,18
In people with T1DM predictable triggers explain many hypoglycaemic events (T1DM),19,20 including skipped meals, strenuous exercise, high insulin doses, chronic renal or liver disease, as well as defective glucose counter-regulation and established hypoglycaemia-associated autonomic failure.21,22,23
Nevertheless, apparently inexplicable hypoglycaemic episodes may be related to incorrect insulin injection techniques including the use of long needles-eventually allowing insulin injection into the subcutaneous muscle tissue—or patients’ incorrect habit to inject insulin into LH nodules.25,26,27
In the series of patients in the Hypos-1 study32,33 a high rate of SHs, NSHs and GV was reported (28%, 70% and 56%, respectively). Accordingly, our data documented an overall 62% Hypos rate, 16.8% in fact being inexplicable and showing a strong correlation with the presence of LH, as well as, with all known associated factors.
The profile of people with diabetes displaying inexplicable Hypos/GV included the presence of LH nodules, as well as, poor metabolic control, high micro-/macrovascular complication rates, longer/larger needles, missed injection site rotation, low socio-economic level and solitary life. The risk for LH to be deemed as responsible for unexplained Hypos or GV rose to 4.38 in the case of simultaneously occurring above mentioned factors and LH.
Conversely, the presence of LH was able to predict about 77.1% of unexplained Hypos/GV. Indeed, factors known to be associated with LH were also present at a high rate in subjects with such phenomena.
However, the remaining 22.9% people without any LH nodules experienced unexplained Hypo/GV too. It might depend on the fact that identifying subtle skin lesions is very difficult so that “no identified LH” is not equivalent at all to “absence of any LH”. In fact, we feel like stressing what was already pointed out by other authors28,31,36: education on injection techniques is still inadequate and diabetes specialists and dedicated nurses should make their best to prevent patients from causing themselves any skin damage through wrong habits.
Our conclusion then is that, in the presence of unexplained Hypos/GV, injection sites should be systematically and thoroughly explored in search of LH areas. Furthermore, appropriate educational activities should be implemented to improve patients’ behaviour, including periodical investigation on their injection habits.
A recent meta-analysis of population based studies42 including 46 publications (n=532, 542) found Hypos to occur quite frequently among people on insulin: mild/moderate events were in fact about 50% with an incidence of 23 episodes per person-year while severe episodes were about 21% with an incidence of 1 event per person-year.
Should we ideally select people with unexplained Hypos from the extremely large number of patients mentioned above, we would deal with an impressive cohort: in that case the systematic utilization of a suitable LH identification protocol followed by patient rehabilitation would be expected to dramatically reduce Hypo prevalence, incidence and health consequences, as well as, the costs related to this frightening, yet neglected, complication of insulin treatment.
Further studies are warranted to assess whether educational activities like those reported in our study may result into a decreased burden of diabetes as for unexplained hypoglycaemia.
CONFLICTS OF INTEREST
The authors declare that they have no conflicts of interest.











