INTRODUCTION
Water is an essential nutrient for many bodily systems and functions, such as regulation of body temperature, lubrication of joints, and optimal cardiovascular function.1 Hydration is especially important in sports and physical activity, due to prodigious perspiration. However, dehydration is a common problem since many athletic individuals do not hydrate properly. Evidence supports the idea that dehydration leads to decrements in physical performance.2,3,4 Regarding cognitive function, many researchers contend that dehydration leads to reduced cognitive function,5,6,7,8,9,10 though the evidence is equivocal.11,12,13,14
One possible reason for the mixed results in the literature is that the methods for testing cognitive function are inconsistent among studies. Because there are many cognitive domains that contribute to cognitive function, some studies focused on specific domains. In many of the studies on athletic populations such as golfers,10 soccer players,15 and jockeys,16 the testing methods were sport-specific to determine the effects of dehydration on the cognitive aspect of their specific sport. Task-specific outcomes are also seen among cognitive tests in military personnel.9,13 While sport-specific testing methods may be ideal for elite athletes, they may not serve recreational or amateur athletes who lack the necessary level of expertise.
Conversely, many studies that tested a wider range of domains chose cognitive tests that may not be ideal for athletic individuals. Though reaction time has been included in many studies to date, researchers often focused on short- and long-term memory5,6 or employed tests that may not translate well to sport, such as perceptive discrimination via visual determination of line length.7 More recently, studies have tested areas such as executive function11 and sustained attention,17,18 but a paucity of data exists in areas such as motor speed and psychomotor speed, which are essential for athletes and athletic individuals. More research needs to be conducted to establish a set of cognitive tests and/or cognitive domains that accurately measure the cognitive functions necessary for athletic performance.
Another reason for the mixed results in the literature is the variety of methods employed to achieve dehydration. Common interventions include: Water deprivation,10,19 passive heat stress,7,11,16 exercise,11,14,20 combined exercise and heat,5,6,12 and comparison of dehydration methods.7,8 Since dehydration can result from various and multiple insults, various dehydration methods must be tested and should reflect the population being studied. In a sport context, most dehydration occurs during exercise in a hot environment; therefore, both exercise and heat should be employed to elicit dehydration to measure cognitive function in athletes of all levels.
Therefore, the purpose of the present study was to determine the effects of mild dehydration in recreational athletes elicited by a dehydration protocol employing both exercise and heat on various domains of cognitive function. Cognitive domains were chosen to be sport-specific and include: Psychomotor speed, reaction time, cognitive flexibility, processing speed, executive function, and motor speed. Our hypothesis was that mild dehydration would cause a decrement in cognitive function in all tests.
METHODS
Subjects
Twenty-seven men (n=14; age: 26.9±4.6 years; height: 176.3±6.6 cm; weight: 79.1±9.0 kg) and women (n=13; age: 27.3±4.3 years; height: 171.0±6.5 cm; weight: 66.5±4.5 kg) participated in the study. All individuals were recreationally trained, defined as exercising two or more times per week for at least one year, as assessed by an exercise status questionnaire. Subjects were free of cardiovascular disease, diabetes, or any other diseases that would exclude them from participation in the study, as assessed by a health history questionnaire. No medications or supplements that could interfere with the study had been consumed for at least one month prior to testing. All protocols for this study were approved by an Institutional Review Board and written informed consent was provided by the subjects prior to testing.
Experimental Design
Subjects arrived to the laboratory in the morning in a euhydrated state after an overnight fast. Subjects were also required to abstain from exercise for 24 hours prior to testing so that fatigue was not a factor. A urine sample was provided and measured for urine specific gravity via refractometry (Atago, Inc., Tokyo, Japan) to ensure hydration status. If the USG value was above 1.025, the subject was considered dehydrated and given 500 ml of water to drink in accordance with ACSM guidelines prior to testing to achieve euhydration at baseline. Urine color, body temperature and skin temperature were measured, followed by cognitive function testing. After baseline testing, subjects performed the dehydration protocol. Subsequently, all tests were repeated.
Dehydration
A combination of active dehydration via treadmill running and passive dehydration via sauna was employed to achieve dehydration. After baseline testing, subjects were required to run on a treadmill (Woodway, Inc., Waukesha, WI, USA) for 30 minutes at 80% maximum heart rate (206.9-(0.67*Age)).21 Afterward, subjects sat in a sauna at 70 °C for 15 minute intervals until 2% body weight loss was attained, which is the value that performance decrements begin to occur.22 Nude, dry body weight was measured between sauna intervals via calibrated scale to ensure that the body weight loss was as close to 2% as possible. When 2% body weight loss was achieved, subjects showered and dried their bodies completely. Nude, dry body weight was measured again and this weight was used as the post-dehydration body weight in the present study.
Measurements
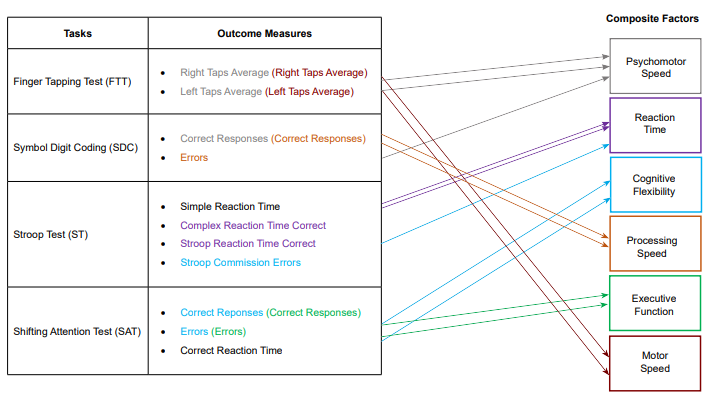
All measurements were taken prior to and immediately following the dehydration protocol. After USG was measured, urine color was measured using a color chart developed by Armstrong and colleagues and validated in young athletes.23,24 Body temperature via digital thermometer (American Diagnostic Corporation, Hauppauge, NY, USA) and skin temperature via forehead infrared thermometer (Telatemp Corp., Anaheim, CA, USA) were measured, immediately followed by cognitive function testing. Subjects sat at a computer and performed a variety of cognitive function tests (CNS Vital Signs Inc., Morrisville, NC, USA), including: Finger tapping test, symbol digit coding, Stroop test, and shifting attention test. The CNS Vital Signs test battery was chosen for its high reliability,25 lack of a learning effect,25 and the application of its tests to sports performance. The finger tapping test requires subjects to tap the space bar with their right hand as many times as they can in ten seconds. The test is repeated with the left hand, and two trials are performed with each hand. The symbol digit coding test involves a serial presentation of symbols and empty boxes below, along with a legend at the top that designates a number to correspond with each possible symbol. Subjects must enter the number that corresponds with the highlighted symbol. The stroop test consists of three parts. In the first part, the words RED, YELLOW, BLUE and GREEN appear at random on the screen and the subject responds as quickly as possible by hitting the space bar (simple reaction time). In the second part, the words RED, YELLOW, BLUE and GREEN appear in color on the screen randomly and the subject responds only when the color matches the word (complex reaction time). In the third part, the words RED, YELLOW, BLUE and GREEN appear in color on the screen randomly and the subject responds only when the color does not match the word (Stroop reaction time). The shifting attention test involves three geometric figures on a screen – one on the top and two on the bottom. The top figure is either a square or a circle. The bottom figures are a square and a circle. The figures are either red or blue (color is randomized). Subjects must match one of the bottom figures with the top figure via arrow keys. The rules of matching are changed randomly (either by color or by shape). Each test started with a practice test to ensure familiarization with the test protocol. The testing room was free of distraction and extraneous noise. A total of 11 outcome variables were collected from each subject, which were then used to calculate 6 cognitive domains (Figure 1).
Figure 1: Cognitive Function Domains Diagram. The Computerized Cognitive Function Test Battery (CNS Vital Signs, Morrisville, NC, USA) is Summarized, Indicating How Tests are Separated into Specific Outcomes, and which Outcomes Contribute to each Cognitive Domain. Colors Correspond with Arrows and Outlines of Cognitive Domains, and Repeated Outcomes Indicate that they Correspond with more than One Domain.
Statistics
All data were analyzed using SPSS software (Version 20.00, SPSS Inc., Chicago, IL, USA). Dependent t-tests were used to calculate time differences between pre- and post-dehydration cognitive function. Since multiple pairwise tests were being performed on the same set of data, a Bonferroni correction was used to adjust the p value. Therefore, significance was set at p≤0.0029.
RESULTS
Mild dehydration was achieved in all subjects (weight lost: 1.51±0.27 kg; percent dehydration: 2.11±0.32%). Significant differences were observed for urine specific gravity values (PRE: 1.008±0.008; POST: 1.015±0.009; p=0.00006) and for urine color (PRE: 3.22±1.27; POST: 4.17±1.30; p=0.018). No significant differences were observed for body temperature (PRE: 36.29±0.21 °C; POST: 36.29±0.19 °C; p>0.05) or for skin temperature (PRE: 34.28±0.60 °C; POST: 34.08±1.30 °C; p>0.05).
All results for cognitive domains and specific test results can be found in Table 1. Significant differences were not demonstrated in any of the six cognitive domains measured: psychomotor speed (PRE: 203.74±23.50; POST: 207.48±27.29; p>0.05); reaction time (PRE: 658.48±63.38ms; POST: 645.67±95.85ms; p>0.05); cognitive flexibility (PRE: 53.22±8.82; POST: 53.93±7.82; p>0.05); processing speed (PRE: 83.11±14.20; POST: 86.04±17.17; p>0.05); executive functioning (PRE:
54.93±8.32; POST: 55.74±7.09; p>0.05); motor speed (PRE: 119.33±13.50; POST: 119.56±16.26; p>0.05). Regarding specific tests, two measurements demonstrated a significant difference from pre to post at p≤0.05, and one measurement demonstrated a significant difference according to the Bonferroni correction (p≤0.0029). The reaction time during the shifting attention test was significantly lower at post-testing, compared to pretesting (PRE: 882.67±126.59ms; POST: 830.00±105.83 ms; p=0.0012).The Stroop reaction time during the Stroop test was significantly lower at post-testing, compared to pretesting (PRE: 712.56±97.52 ms; POST: 671.63±97.21 ms; p=0.02), but was not significant when the Bonferroni correction was applied. No significant changes were observed in any other measurements during specific tests.
| Table 1: Summary Data for Cognitive Function Domains and Specific Test Results before and after Dehydration. |
| |
|
Mean
|
SD |
Mean Difference |
p Value |
Effect Size (d)
|
|
Psychomotor speed
|
Pre |
203.74 |
23.50 |
3.74 |
0.16 |
0.14 |
| Post |
207.48 |
27.29 |
|
|
|
|
Reaction time†
|
Pre |
658.48 |
63.38 |
-12.81 |
0.33 |
0.24 |
| Post |
645.67 |
95.85
|
|
|
|
|
Cognitive flexibility
|
Pre |
53.22 |
8.82 |
0.70 |
0.49 |
0.36 |
| Post |
53.93 |
7.82
|
|
|
|
|
Processing speed
|
Pre |
83.11 |
14.20 |
2.93 |
0.13 |
0.54 |
| Post |
86.04 |
17.17
|
|
|
|
|
Executive functioning
|
Pre |
54.93 |
8.32 |
0.81 |
0.45 |
0.47 |
| Post |
55.74 |
7.09
|
|
|
|
|
Motor speed
|
Pre |
119.33 |
13.50 |
0.22 |
0.89 |
0.04 |
| Post |
119.56 |
16.26
|
|
|
|
|
Finger tapping test: right taps average
|
Pre |
62.63 |
7.60 |
-0.48 |
0.63 |
0.22 |
| Post |
62.15 |
10.38
|
|
|
|
|
Finger tapping test: left taps average
|
Pre |
56.33 |
7.18 |
1.04 |
0.27 |
0.72 |
| Post |
57.37 |
7.11
|
|
|
|
|
Symbol digit coding: correct
|
Pre |
84.41 |
14.69 |
2.52 |
0.19 |
0.44 |
| Post |
86.93 |
17.58
|
|
|
|
|
Symbol digit coding: errors†
|
Pre |
1.30 |
1.81 |
0.22 |
0.54 |
2.59 |
| Post |
1.52 |
1.31
|
|
|
|
|
Stroop: simple reaction time†
|
Pre |
345.93 |
41.87 |
-6.70 |
0.45 |
0.25 |
| Post |
339.22 |
49.02
|
|
|
|
|
Stroop: complex reaction time†
|
Pre |
604.78 |
69.24 |
14.52 |
0.46 |
0.22 |
| Post |
619.30 |
123.46
|
|
|
|
|
Stroop: Stroop reaction time†
|
Pre |
712.56 |
97.52 |
-40.93 |
0.02* |
0.55 |
| Post |
671.63 |
97.21
|
|
|
|
|
Stroop: commission errors†
|
Pre |
1.67 |
1.57 |
0.15 |
0.75 |
0.99 |
| Post |
1.81 |
2.48
|
|
|
|
|
Shifting attention test: correct
|
Pre |
57.74 |
6.65 |
1.48 |
0.07 |
1.29 |
| Post |
59.22 |
5.77
|
|
|
|
|
Shifting attention test: errors†
|
Pre |
2.81 |
2.56 |
0.67 |
0.08 |
3.75 |
| Post |
3.48 |
2.05
|
|
|
|
|
Shifting attention test: reaction time†
|
Pre |
882.67 |
126.59 |
-52.67 |
0.00121** |
0.53 |
| Post |
830.00 |
105.83 |
|
|
|
| *indicates post significantly different from pre at p≤0.05. **indicates post significantly different from pre at p≤0.0029. †indicates tests in which a lower score is better. |
DISCUSSION
In contrast to our hypothesis, the main findings of the present study indicated that Stroop reaction time during the Stroop test and reaction time during the shifting attention test were enhanced by a sport-specific dehydration protocol employing both heat and exercise, and reaction time during the shifting attention test was significantly different even after the Bonferroni correction was applied. Previous studies have generally found that dehydration caused a decrement in cognitive function5,6,7,8,9,10 or that no significant changes occurred.13-15,18,19,26 However, three studies did demonstrate cognitive improvement after dehydration.11,12,13 It is possible that the differences among cognitive function assessment tools and domains contributed to the discrepancy in findings. For example, Tomporowski and associates11 employed a category-switching task developed by Kramer27 that is very similar to the shifting attention test in the present study. Reaction times were faster after exercise in the Tomporowski study, regardless of whether subjects were 3% dehydrated or if fluid replacement kept them hydrated, which supports the faster reaction times during the shifting attention test in the present study. Also, errors significantly increased after exercise during the category-switching task when the response required the subject to switch categories, i.e. when the task became more complex, as opposed to a non-switching response.11 Similarly, there was a trend observed for errors (p=0.08) on the shifting attention test in the present study. Therefore, consistency in testing methods is essential in order to compare results among studies.
In other studies that tested for reaction time, results were either positive or not significant. Leibowitz et al12 induced either 2.5% or 5% dehydration using exercise performed in a heat chamber, which improved reaction time in response to peripheral stimuli but no change in response to central stimuli. In a marksmanship test in volunteer soldiers, response time between target presentation and firing of the rifle improved after exercise with no effect from 2.6% dehydration.13 Cullen et al16 observed no effect of 4% dehydration on choice reaction time in jockeys, which was supported in other studies employing water deprivation to achieve 1.5% and unreported dehydration, respectively.10,19 Though the literature is not conclusive, it is interesting to note that most of the studies that demonstrated cognitive decrements did not test for reaction time.5,6,9,10 In the study by Cian and associates where cognitive decrements were observed after 2.7% dehydration and choice reaction time was measured, cognitive function worsened in perceptual discrimination and short-term and long-term memory, but no changes were observed in choice reaction time.7 Therefore, it seems plausible that choice reaction time is a cognitive domain that is insensitive to dehydration or is improved in some cases of exercise-induced dehydration.
Since there are a variety of cognitive domains that contribute to cognitive function, it is important to test domains that are specific to the population and the outcomes of interest. In healthy, active individuals, there are many possible outcomes of interest, such as quality of life, decreased risk for chronic disease, and improvements in work output, school performance, and sports performance. Cognitive domains such as short- and long-term memory may be more applicable to quality of life and school performance, whereas choice reaction time and motor skills may be more applicable to sports performance. The domains chosen for the present study were derived from previous studies in athletes to translate to sports performance. Bandelow et al15 tested soccer players on the same motor skills test (finger tapping test) and with a choice reaction time test that tested the same cognitive domain as the complex reaction time section of the Stroop test.15 Triathletes were tested for simple and choice reaction time, which are tested in the Stroop test.28 Track and field athletes were tested on a variety of executive and non-executive tasks to determine the effects of exercise on executive function, which was tested via the shifting attention test in the present study.29
Interestingly, Stroop reaction time was the only reaction time test measured by the Stroop test that demonstrated improvements, though it is the most complex. Stroop reaction time measures the responses in the third part of the Stroop test, where a response must be given when the color of the word does not match the word written. This was a novel finding since it suggested that the subjects’ ability to respond to rapidly changing and increasingly complex sets of directions was improved, and other researchers have suggested that an attenuation of exerciseinduced cognitive benefits occurs as complexity increases, because more errors were made in tasks that required subjects to shift decision-making rules.11 This was not the case in the present study, in which the most robust effect was observed from the shifting attention test, which was significant even after the Bonferroni correction was applied.
Another area of discrepancy in the literature is the method of dehydration. Various methods of dehydration are employed in studies, and dehydration methods should reflect how dehydration occurs in specific populations. For example, fluid deprivation is probably the most common method by which dehydration occurs in everyday life. In most athletic situations involving dehydration, both heat and exercise play a role. In military, tactical and sport contexts, intense exercise is often performed in hot, humid conditions. Therefore, dehydration protocols may be more applicable to sport when heat and exercise are both employed, even though studies have not demonstrated significant differences between deprivation methods.7,8 Since exercise was employed as a method of dehydration in the present study, the cognitive improvements may have been caused by the exercise and not the dehydration. Multiple studies have demonstrated improved cognitive function after exercise.9,20,30,31,32 For example, Nanda et al30 elicited improvements in memory, reasoning, and planning after 30 minutes of cycling at 70% of heart rate reserve.30 Hillman and associates found that amplitude of event-related brain potential increased after 30 minutes of treadmill running,20 which involved a similar exercise bout as employed by the present study. One possible mechanism is that exercise increases blood flow to the brain, which has been linked to improvements in cognitive function.33 The exercise-induced dehydration may have elicited positive cholinergic effects, as demonstrated in an animal study by Fordyce et al.34 Though exercise may be contributing to the cognitive benefits, it is important to note that not all exercise-induced dehydration studies demonstrated improvements.5,33
There are limitations regarding interpretation of the findings of the present study. First, the combination of exercise and heat stress in the dehydration protocol limits the ability to determine whether exercise or dehydration is causing the improvements, though employing both dehydration methods was important to apply the dehydration to sport contexts. Second, our null results and trends may be non-significant due to small data set size. Third, the subjects were athletic, active individuals, but were not collegiate or professional athletes. Therefore, it is difficult to determine whether the findings would apply to highly athletic men and women. Future studies investigating dehydration and cognitive function need to enlist elite athletes in various sports to support the use of sport-specific cognitive testing methods and dehydration protocols.
CONCLUSION
In summary, mild dehydration caused by both exercise and heat exposure enhanced Stroop reaction time during the Stroop test. Second, reaction time during the shifting attention test was significantly different after dehydration, even after the Bonferroni correction was applied. These findings have military, tactical, and sport applications, in which athletes and athletic individuals are often performing at various levels of dehydration caused by both heat and exercise, and require optimal cognitive functioning in multiple, specific areas. Also, weight-class athletes may employ both exercise and sauna to achieve weight loss, which may induce quicker reaction times when the dehydration is mild. More research is needed to elucidate the precise areas of cognitive function that improve or decline as a result of mild dehydration, and whether elite athletes will demonstrate the same improvements as athletic individuals.
ACKNOWLEDGEMENTS
We would like to thank MusclePharm Inc., for funding for this study. Citation of commercial organizations or products does not constitute endorsement by the authors.
CONFLICTS OF INTEREST
The authors declared that they have no conflicts of interest.






