INTRODUCTION
Merkel cell carcinoma (MCC) is an uncommon neuroendocrine neoplasia occurring in people over 55-years-old and more frequently in the males.1 MCC originates in the cells of the basal epidermal layer that are involved in proprioception.2 From the clinical perspective, it presents as a violaceous and painless erythematous nodule-rarely ulcerated in the skin. Excessive exposure to ultraviolet light is considered to be the main risk factor, and people with immune suppression are thirteen times more likely to develop it.3 The exact pathogenesis is uncertain, but associated with Polyomavirus.4
It should be noted that MCC is an aggressive tumor, sensitive to chemotherapy and radiation therapy. Local relapses are common and regional and distant metastasis are frequent.5
However, testicular metastases are extremely rare, and less than ten cases have been reported in the literature to date.6
CASE REPORT
The patient was a 65-year-old male with a past history of high blood pressure, dyslipidemia, pancytopenia, and Chagas disease. He had been diagnosed in 2016 with MCC in the right thigh and a history of inguinal iliac resection with safety margins, chemotherapy and radiation therapy. He consulted because of an increase in size of the right testicle with concomitant pain of 1-month duration.
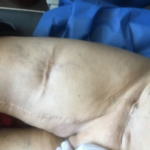
During the physical exama surgical scar covering upper third of right leg up to inguinal region was noted. The enlarged scrotum at the expense of a right testicle with painful in duration, about three centimeters that makes it difficult to palpate the epididymis and does not appear to be attached to the skin. The left testicle was normal (Figure 1).
Figure 1. MCC Surgical Scar Onrightthigh
Scrotal ultrasound reported an increased volume of the right testicle with multiple hypoechoic nodular solid lesions compatible with neoplasia. The left testicle had a normal volume, but showed similar nodular lesions in smaller numbers. There was a bilateral positive bilateral Doppler that ruled out testicular ischemia.
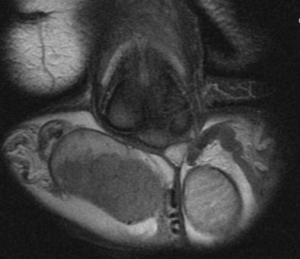
Laboratory studies showed pancytopenia and negative testicular tumor markers. Computed tomography (CT) was performed without showing dissemination of disease. Magnetic resonance imaging (MRI) reported a marked increase in the size of the right testicle due to nodular lesions with hypo intensity in T1 and T2. The largest lesion was thirty millimeters in diameter and showed diffuse enhancement after contrast. The left testicle size, morphology and signal intensity were all normal (Figure 2).
Figure 2. Coronal View of Genitals in T2 Sequence
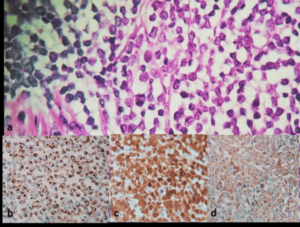
Right radical or chiectomy was performed. The deferred histopathology study showed a deformed testicle due to various nodule-shaped prominences with preservation of the tunica albuginea. Section of the specimen showed light brown and brownish lesion of increased consistency that occupies almost the whole test is with extension to the tunica albuginea. Immuno-histochemistry showed positive results for CK 20, synaptophysin, chromogranin and enolase; and a negative result for CK 7. This pattern was consistent with a diagnosis of testicular MCC metastasis. The patient started adjuvant immunotherapy after the diagnosis (Figure 3).
Figure 3. a. Atypical Neoplastic Proliferation, Made-up of Nests with Uniform Blue Rounded with Granular
Salt and Pepper Chromatin and Little Cytoplasm. b. CK 20 Positive. c. Enolase Positive. d. Synaptophysin Positive
DISCUSSION AND CONCLUSION
Merkel cell carcinoma was described by Toker in 1972 as trabecular carcinoma and is also known as Toker’s Tumor.7 Remarkably, there are few cases registered in Argentina.8,9,10,11 The disease occurs most commonly in people older than 50 and there is a view that the testicle is a sanctuary site for this carcinoma because the blood-test is barrier prevents the eradication of tumor cells by adjuvant chemotherapy.12,13 It must also be noted that the patient also underwent inguinal lymphadenectomy and radiotherapy ipsilateral to the tumor localization, so the clinical history could favor the development of metastasis in the testicle.
Immuno-histochemical analysis is essential to differentiate MCC from other tumors. Furthermore, MCC is associated with positive staining for CK 20, CAM 5.2, CK AE1/AE3 and neuroendocrine markers such as synaptophysin and chromogranin. By way of contrast, metastasis from small lung cell carcinoma is positive for CK7; lymphoma expresses CD45 and melanoma metastases stain positive for S100, HMB-45 Melan-A.14
Merkel cell polyomavirus (MCPyV), usually does not cause disease, but can be integrated into the host DNA and cause MCC. MCPyV is not commonly observed in other malignancies and is therefore also used as a marker to support a diagnosis of MCC. However, its value as a prognostic marker, is not yet clear.15
The patient also had immune suppression secondary to myelodysplasia which increases the risk of MCC thirteen-fold. Sun exposure and mutations in the p53 gene variants are additional risk factors.16
Treatment of MCC by stage, according to the American Joint Cancer Committee in its eighth edition, states that the following should be performed: 1) Stage I and II excision; 2) Stage III (Nodal Disease) extensive lymphadenectomy excision with radiation therapy of the primary tumor or adjuvant chemotherapy versus immunotherapy versus targeted therapy; and 3) Stage IV chemotherapy/immunotherapy. In the event of relapse, multidisciplinary treatment will be considered,17 as presented in the reported case where the patient received surgical treatment, chemotherapy and radiation therapy, followed by immune-therapy always with multidisciplinary management. The survival of patients varies from 62.8% in stage I to 13.5% in stage IV.18
To summarize, the role of immuno-histochemistry is essential in the diagnosis of possible testicular metastasis in patients with a past history of MCC. Strict multidisciplinary management is useful in these cases and with advanced stage disease immunotherapy appears to be promising. However, there are very few cases described in the literature to date.
CONSENT
The authors have received written informed consent from the patient.
CONFLICTS OF INTEREST
The authors declare that they have no conflicts of interest.








