INTRODUCTION
Measles continues to cause death and severe complications including pneumonia, croup, encephalitis, blindness and otitis media in children and it remains one of the most important causes of child morbidity and mortality globally.1 Following an infection, the virus can persist for a long time in the body contributing to the development of lifelong immunity.2 According to the World Health Organization (WHO),3 measles can be prevented readily by vaccination, reaching and maintaining high immunization coverage in a population. Thus the organization recommends immunization against measles for all susceptible children and adults for whom measles vaccination is not contraindicated in order to reach and maintain a population herd immunity of at least 90% in all second administrative levels, a prerequisite for prevention of measles epidemics. In addition, the World Health Organization (WHO) recommends two doses of measles for all children with the first dose in infancy and the second dose may occur either at a scheduled age through routine services or periodically through Supplementary Immunization Activities (SIAs) or mass campaigns targeting at least 95% coverage. The organization has also developed the Measles Strategic Planning (MSP) tool to harness routinely available data to estimate effectiveness and cost effectiveness of vaccination strategies.4 The vision of the Global measles and rubella strategic plan: 2012-20205 is to achieve and maintain a world without measles, rubella and Congenital Rubella Syndrome (CRS) or measles eradication is defined as the world-wide interruption of transmission of the virus, and represents the sum of successful elimination efforts in all countries and regions.6
South Sudan revitalized measles programme after the Comprehensive Peace Agreement (CPA) in 2005 with a catch-up campaign conducted in 2005-2007 recording post campaign evaluation coverage of 74%. The catch-up campaign was followed by Accelerated Child Survival Initiatives (ACSI) in 2009 and a follow-up campaign in 2012 which recorded administrative coverage of 62% and 92% respectively. The country’s measles elimination strategy from 2014-2020 was developed in 2013 which sets out objectives in line with the regional and global strategic goals. In April 2014, another follow-up campaign was implemented in seven states, and partially in 25 counties in the three conflicts affected states of Jonglei, Unity and Upper Nile. However, over the period of 2005-2015, laboratory confirmed measles outbreaks occur every year, and are responded to with selective vaccination with limited geographic scope usually within the affected county or its entirety but not in bordering counties at risk. Routine immunization is mostly conducted by NGOs, yet it is limited to the functional heath facilities of which an estimated 32% provide mainly fixed vaccination services. Since 44% of the population have access to functioning health facilities, the Ministry of Health (MoH) with the support of partners conducts three rounds of outreach sessions in a campaign mode in a year. The conflict that started in December 2013 limits access to vaccination campaigns. Thus, the current measles vaccination programme is limited to routine vaccination mostly implemented at functional health facilities and outbreak campaign response. Surveillance remains largely passive under the Integrated Disease Surveillance and Response system currently under development. However, a Case-based surveillance was established in October 2011 in a parallel mode setting the stage for the elimination agenda.
Even though routine immunization is known to be the cornerstone in eliminating vaccine preventable diseases such as measles, fragile routine immunization system coupled with the complexity of emergencies in the context of South Sudan have led to reliance on mass campaigns to reach elimination targets. However, the effectiveness of follow-up campaigns as compared with outbreak responses in reducing the burden of measles in the population remains uncertain. The aim of this study is to determine the effectiveness of the targeted measles outbreak vaccinations currently practiced in South Sudan for measles elimination.
METHODS
We used time trend analysis of secondary data sources for routine and supplementary measles vaccination, and an analytical cross-sectional design on the dataset of case-based surveillance on measles in South Sudan from 2011-September 2015 to examine the risk of measles in the population over the period.
The exposure factor was evidence of history of vaccination against the outcome of IgM test results for measles.
Data Sources
The WHO/Unicef Joint Report estimates for 2011, 2013-2014, Expanded Programme on Immunization (EPI) Coverage Survey for 2012, and Administrative data source for 2015 were used for routine immunization coverage for measles. Supplementary Immunization data were sourced from reports from measles follow-up, preventive and outbreaks vaccination campaigns over the period 2011-2015.
The centralized measles case-based surveillance database hold investigated measles cases from all reporting health facilities. Data on measles cases are captured on a generic IDSR case-based form containing all relevant variables: epidemiological numbers, age, sex, residence, and date of onset of rash as well as date and doses of previous vaccinations, dates of notification, and results of samples. The database is on Microsoft Access Database (mdb) platform designed with Epi Info 3.5.0.
Serology
Whole blood venipuncture specimens were obtained from suspected measles cases in health facilities, kept unfrozen at 4-8 °C for 24 hours until complete retraction of clot and serum is separated or; allowed to clot and centrifuged at 100 g for 10 minutes to separate the serum. The separated serum was transferred into serum tubes and stored at 20 °C until transported in a specimen carrier with ice packs, to the National Public Health Reference Laboratory and kept at 20 °C until tested. WHO validated Dade Behring kits were used to perform measles indirect IgM test.
Measles cases were defined by laboratory confirmation of IgM presence for measles.
Data Analysis
We mapped the measles routine immunization and SIA coverage data over the years 2011-2015. The measles line list was extracted from the Measles Case based dataset and analyzed using EPI info version 7 and Statistical Package for Social Sciences (SPSS) version 20. Data were cleaned and validated with reference to filed hard copies of the case investigation forms.
We excluded cases without serum samples, as well as cases with samples but with date of previous measles vaccination within 28 days before onset of rash. In addition, epidemiologically linked and compatible cases were also excluded in the analysis in terms of comparing with IgM negative cases in order to control bias resulting from discretional definition of such cases.
The trends of measles cases were mapped with types of measles SIAs over time in an epidemiological curve. Cases with evidence of vaccination and IgM +ve test results for measles were recorded 1 and the absence of both variables, 0. Measles cases with evidence of no history of vaccination were used as referent and binary logistic analysis used to estimate the odds of measles IgM +ve cases accounting for variability at 95% confidence interval for each year. Interactive logistic regression was also applied to adjust separately for influences of age and cases with serum samples but with unknown vaccination status on the odds ratio of measles among vaccinated population over time. Estimates of odds and Mcffaden’s pseudo–R square were made. The analysis to some extent controlled for the effects of recall or investigation biases that informed records on vaccination status, but absence of data on corresponding vaccination intervention to doses as well as co-morbidity were limitations hence, uncontrolled and therefore limited estimation of vaccine effectiveness. Notwithstanding, the assumption made was that vaccination doses where largely related to major interventions (follow-up campaigns or outbreak response vaccinations) within the year.
RESULTS
The WHO and UNICEF joint estimate puts routine immunization coverage for third dose Diphtheria Pertussis Tetanus (DPT3) at 60.0%, 65.0% and 47.0% for 2011, 2013 and 2014 respectively. The EPI Coverage survey in 2012 estimated DPT3 coverage of 45.9%. The administrative coverage for routine immunization in 2015 was 52.1%.
Administrative coverages of selective measles outbreak response vaccinations in 2011 and 2013 were 92.0% and 90.2% respectively. Post campaign coverage survey after the Follow-up campaigns conducted in 2012 and 2014 recorded 92.8% by figure mark and 88.0% evidenced by card respectively (Table 1).
| Table 1: Routine immunization coverage and SIAs. |
|
Year
|
Routine Immunization (%) |
Supplementary Immunization Activities (%) |
| 2011 |
60.0b |
92.0
|
|
2012
|
45.9a |
92.8c |
| 2013 |
65.0b |
90.2
|
|
2014
|
47.0b |
88.0c |
| 2015 |
52.1d |
|
| aEPI Coverage Survey (2011)
bJoint WHO UNICEF estimate
cPCE Coverage from F-up campaign
dAnnualised Routine Immunization |
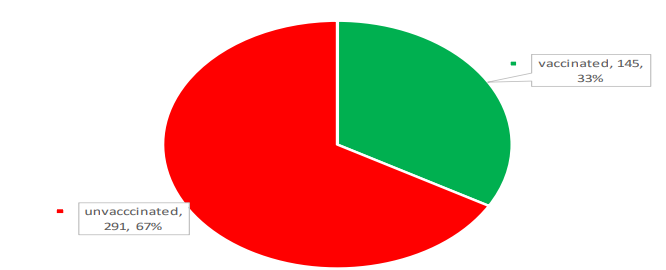
Out of the 1087 suspected measles cases investigated from 2011-2015, 436 had evidence of known vaccination status, of which 291 representing 66.7% (95% CI 62.8-71.1) had received no dose of measles vaccine as against 145, forming 33.3% (95% CI 28.9, 37.9) with evidence of receipt of a dose or more
(Figure 1).
Figure 1: Proportional distribution of vaccination status of investigated measles cases 2011-2015, South Sudan (n=436).
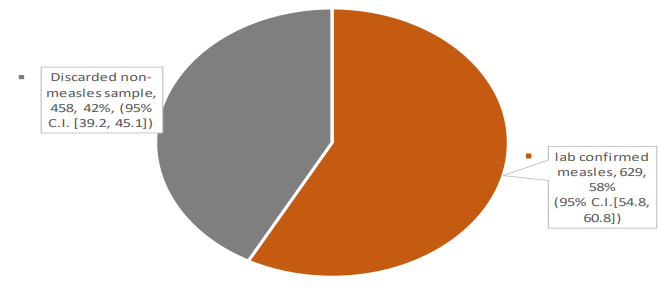
Of the total cases investigated, 629 (57.9% (95% CI 54.8, 60.8) were laboratory confirmed measles cases and 458 (42.1%) discarded as non-measles cases as shown in Figure 2. However of the 436 cases with evidence of known vaccination status, 62.1% (95% CI 57.4, 66.7) were lab confirmed measles cases (Immunoglobulin M (IgM) positive), and the rest, 37.8% (95% CI 33.3, 42.6) non measles cases (IgM negative) (Figure 2).
Figure 2: Proportional distribution of laboratory result of investigated measles cases, 2011-2015, South Sudan.
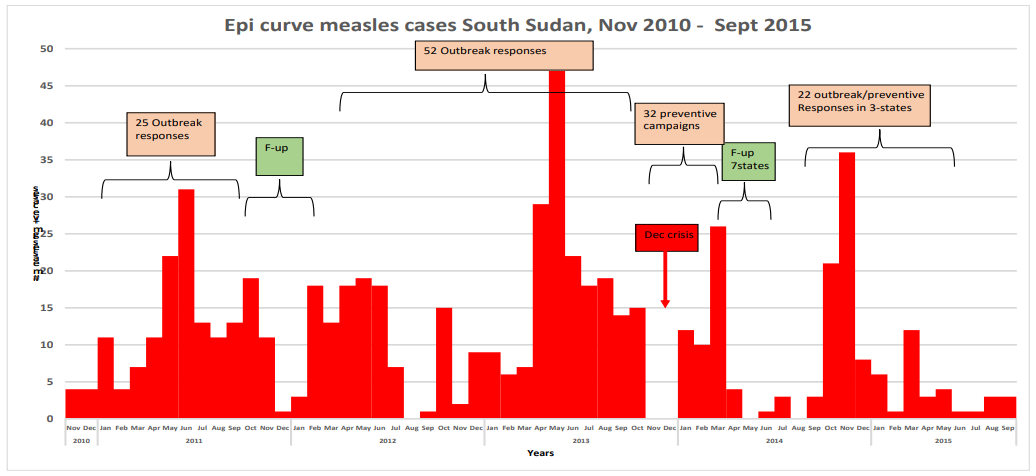
Measles cases have multiple peaks in a year. In 2013, peaks were observed in January with 11 cases, 32 cases in May and 18 cases in October. In 2012 four peaks were observed in February (17 cases), May (18 cases) October (15 cases) and 8 cases in December. In 2013 the first peak of 8 cases in January rose sharply to 48 cases in May and declined to 15 cases in October. In the same year, 2013 cases were not reported in November and December. The three peaks in 2014 were recorded in January (12 cases), March 26 cases and 36 cases in November. In 2015 a sharp rise of cases were noted in March (12 cases) but declined to 2 cases in August as detailed in Figure 3.
Figure 3: Epi curve of measles cases with history of vaccination 2010-2015.
The proportion of confirmed measles cases with history of vaccination decreased from 24.3% in 2011 to 14.2% in 2012 but with a sharp rise to 61.9% in 2014 and a drop to 21.4% in 2015 as shown in Table 1 below. Even though measles vaccination reduced the risk of the disease by 30% (odds 0.7; 95% CI 0.4-1.0) from 2011-2015, a trend analysis showed that the likelihood of reduction of measles burden due to measles vaccination varied per year, but a higher reduction of risk were observed in years (2012 (odds 1.06 (95% CI 0.32-3.55) and 2015 (odds 0.05 (95% CI <0.01-0.35) preceded by follow-up campaigns (Table 2).
| Table 2: Trend analysis of odds of measles cases among vaccinated population. |
|
Year
|
Total number IgM +ve |
Vaccinated |
| % IgM +ve |
Odds |
95% CI |
| 2011-2015 |
145 |
29.9 |
0.70 |
0.4, 1.0
|
|
2011
|
74 |
24.3 |
1.99 |
0.67, 5.88 |
| 2012 |
56 |
14.2 |
1.06 |
0.32, 3.55
|
|
2013
|
85 |
30.5 |
1.45 |
0.55, 4.02 |
| 2014 |
42 |
61.9 |
0.81 |
0.32, 2.02
|
|
2015
|
14 |
21.4 |
0.05 |
0.00, 0.35
|
Adjusting for samples with unknown vaccination status, the odds of measles among samples with known vaccination status increased in 2012 and 2013 but sharply dropped in 2014 to 2015. The decrease in 2012 and 2014 cannot be explained by the vaccination, however in 2015, 45% of the unit change of reduced risk to measles infection can be explained by the vaccination status of the sample (OR 0.01 (95% CI 0.01-0.35) R2 is 0.45) (Table 3).
| Table 3: Odds of IgM +ve per year adjusted of unknown vaccination status and age. |
|
Year
|
OR adjusted for unknown vaccination status |
OR adjusted for age |
| OR [95% CI] |
R2 |
OR (95% CI] |
R2
|
|
2011
|
0.6 [0.30, 1.56] |
0.09 |
0.90 [0.81, 0.95]* |
0.10 |
| 2012 |
1.1 [0.30, 3.51] |
0.00 |
0.90 [0.80, 1.00] |
0.04
|
|
2013
|
1.5 [0.51, 3.80] |
0.08 |
1.01 [0.91, 1.12] |
0.19 |
| 2014 |
0.8 [0.30, 2.10] |
0.00 |
0.99[0.94, 1.04] |
0.00
|
|
2015
|
0.01 [0.01, 0.35]* |
0.45 |
0.98 [0.71, 1.23] |
0.47
|
| *p<0.05 |
The level of likelihood of reduction of measles in the vaccinated population when adjusted for age was generally less than 10% from 2011-2012. A higher percentage of the phenomenon in 2013 (19%) and 2015 (45%) could be explained by a unit change of age of the population, however in 2014 the unequivocal risk of measles between vaccinated and unvaccinated population cannot be explained by the age between the groups (R2=0.00). This notwithstanding there is no significant difference between the levels of likelihood of reduction of measles among the vaccinated population from 2011 to 2012.
DISCUSSIONS
Globally measles control and elimination strategies have made tremendous impact on childhood morbidity and mortality.3,4 Optimum benefits are achieved following high quality and high coverage through routine immunization, follow-up campaigns and outbreak vaccinations as well as effective case management integrated with appropriate administration of doses of Vitamin A.3 Sustained high routine immunization coverage for 1st and 2nd doses of Measles Containing Vaccines (MCV) is known to enhance population herd immunity of children thereby reducing levels of population susceptibility.4,6 South Sudan measles elimination strategy which was preceded by a control strategy during the 2005 CPA period formed the foundation of formalizing EPI structures which was underdeveloped and therefore, has remain very fragile with sub-optimal outcomes. The two years phased catch-up campaign in 2005-2007 in addition to the accelerated child survival intervention in 2009 recorded below 70% coverage. Yet, gains could not be sustained in the context of the weak routine immunization system which recorded estimates below 70% at both national and sub-national levels since 2011 to 2015. The low performance may also be accounted for by the multiple populations mix following the 2011 independence resulting in 2.7 million influx of returnees and refugees from neighboring countries thereby raising susceptibility levels and precipitating rapid transmission of the virus which has been further worsened by the crisis in December 2013 that has caused the displacement of over 1.6 million people.6 The record of low coverage for measles intervention in deprived settings and the resurgence of measles outbreaks have been recorded in parts of Africa and parts of East Asia7,8 reaffirming the need for strengthened health systems for disease control and elimination.
Measles follow-up campaign provides opportunity to reduce high levels of susceptible population and further enhance herd immunity through the provision of opportunity for a second MCV dose.2,9 The acceptable coverage for measles follow-up campaigns is at least 95% preferably verified by card following which period of outbreaks lengthens for 1-3years and frequency reduced.6 During outbreaks selective vaccination coupled with enhanced routine immunization and active surveillance are recommended.1,4,6 In this study, follow up campaigns in South Sudan usually were preceded by an average of 20 lab confirmed outbreak response vaccinations per year and was conducted in 2 years interval. SIAs coverages were however sub-optimal due to multiplicity of systemic factors including limited number of qualified persons to administer the vaccine, barriers in terms of geographic access as well as limited capacities of implementing partners. Additional barriers were limited cold chain infrastructure and inadequate management of existing ones to ensure vaccine efficacy both in routine and follow-up campaigns. All follow-up campaigns were therefore phased, allowing for high number of missed target population. Consequently, the trend of peaks of measles cases, affirm the short interval of build-up of susceptibles. Similar trends have been observed in many difficult settings in Africa and South America were follow-up campaigns and intervals of resurgence of multiple outbreaks occurred in less than 2 years.10,11 Implementation of high quality follow-up campaigns in low resourced settings therefore remains a challenge.
Measles vaccine is cost effective in mitigating the burden of the disease,1,6,12,13 however it is among the most heat sensitive vaccines. Incidences of measles have seen a drastic decline as evidenced in many settings including developing countries.5,14 It is established that approximately 85% and 95% of population receiving measles first and second dose respectively could reach optimum seroconversion rates against the disease.6,14,15 South Sudan has an open vial policy for measles. Our findings showed that in South Sudan, high percentage (29.9% (95% CI 22.5, 37.4) of measles cases with history of previous doses of MCV were laboratory confirmed discounting for MCV doses given within 28 days before onset of rash. We also observed that yearly trends of proportions of laboratory confirmed measles cases among vaccinated cases peaked in years were outbreak vaccinations were conducted but reduced in years following follow-up campaigns. The high percentage of measles cases among the vaccinated population could be attributed to vaccine failure due to poor cold chain, vaccine handling and administrative practices, noting the weak infrastructure and human resources capacities in the country. In recent times, similar factors have accounted for the occurrence of outbreaks in both developing and underdeveloped countries.10,11,16,17,18,19 Comparing the risk of measles among vaccinated population over time was limited by possible recall bias since vaccination status of cases was not evidenced by card and detailed history of vaccine handling. Therefore, the extent of variation of trends that could be attributed to vaccine failure may be deficient but possibly cannot be discounted considering the weak health system.
The burden of mortality and morbidity due to measles and accompanying socio-economic cost have been well acknowledged globally to have declined significantly through vaccinations.5,12,18 In this study, we found that from 2011-2015 a unit change of doses of measles vaccines administered into the population reduced the risk of measles by 30%. A yearly analysis however indicated excess risk due to vaccine administration usually when only outbreak response were conducted in most part of the year as reflected in the increasing burden of the disease among vaccinated population in 2011, 2013 and 2014. Poor quality outbreak response largely due to improper microplanning, poor cold chain practices and inadequate training are major contributors to ineffectiveness of responses. The overdependence on non-state actors as implementing partners within the context of inadequate legal, regulatory and coordination regime could negatively affect proper supervision and quality assurance monitoring mechanisms for interventions including measles outbreak response. We also noted that the odds of measles in the vaccinated population reduced in a year following the follow-up campaigns as evidenced in 2012 and 2015; however, the level of reduction overlaps with the 95% confidence interval constructs for years where only outbreak response vaccinations were conducted. The follow-up campaigns therefore had higher likelihood of measles risk reduction due to the wider geographic coverage unlike selective vaccination following measles outbreaks.
CONCLUSIONS
Targeted measles outbreak vaccinations generally reduced the burden of measles however the extent of reduction is more reflected in a year following a mass measles campaign as compared to selective vaccination following measles outbreaks. Thus measles follow-up campaigns are necessary for sustained measles control and elimination.
Strengthening routine immunization and case-based surveillance in South Sudan could results in a marked sustainability of achieving measles elimination targets over time; however, due to the current crisis and associated high displaced populations and weakened health infrastructure, follow-up campaigns could make a significant impact on the population in reducing the disease burden towards the global measles elimination goals.
ACKNOWLEDGEMENTS
We thank Boniface Imbani, Melisachew Andane, Taban Musa, Sisto Angelo and Patrick Iranya for the assistance during the investigation.
CONFLICTS OF INTEREST: None.








