1. Bracarda s, Ruggeri EM, Monti M, et al. Early detection, prevention and management of cutaneous adverse events due to sorafenib: Recommendations from the sorafenib working group. Crit Rev Oncol Hematol. 2012; 82: 378-386. doi: 10.1016/j. critrevonc.2011.08.005
2. Wilhelm SM, Adnane L, Newell P. Preclinical overview of sorafenib, a multikinase inhibitor that targets both Raf and VEGF and PDGF receptor tyrosine kinase signaling. Mol Cancer Ther. 2008; 7: 3129-3140. doi: 10.1158/1535-7163.MCT-08-0013
3. Capdevila J, Iglesias L, Halperin I, et al. Sorafenib in metastatic thyroid cancer. Endocrine Related cancer. 2012; 19: 209-216.doi: 10.1530/ERC-11-0351
4. Llovet JM, Ricci S, Mazzaferro V, et al. SHARP Investigators Study Group. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008; 359: 378-390. doi: 10.1056/NEJMoa0708857
5. Hutson TE, Bellmunt J, Porta C, et al. Long-term safety with sorafenib in advanced renal cell carcinoma:follow-up of patients from phase III TARGET. Eur J Cancer. 2010; 46: 2432-24340. doi: 10.1016/j.ejca.2010.06.121
6. Naito S, Tsukamoto T, Murai M, Fukino K, Akaza H. Overall survival and good tolerability of long-term use of sorafenib after cytokine treatment: final results of a phase II trial of sorafenib in Japanese patients with metastatic renal cell carcinoma. BJU Int. 2011; 108: 1813-1819. doi: 10.1111/j.1464-410X.2011.10281.x
7. Porta C, Paglino C, Imarisio I, Bonomi L. Uncovering Pandora’s vase: The growing problem of new toxicities from novel anticancer agents.The case of sorafenib and sunitinib. Clin Exp Med. 2007; 7: 127-134. doi: 10.1007/s10238-007-0145-8
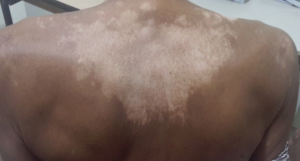
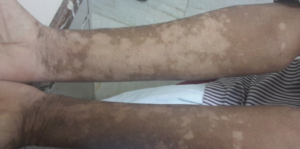
8. Hussain SZ, Asghar A, Ikram M, Islam N. Development of skin hypopigmentation in a patient with metastatic papillary carcinoma thyroid treated with Sorafenib. BMC Endocr Disord. 2013. 13: 29. doi: 10.1186/1472-6823-13-29
9. Lacouture ME, Wu S, Robert C. Evolving strategies for the management of hand-foot reaction associated with the multitargeted kinase inhibitors sorafenib and sunitinib. Oncologist. 2008; 13: 1001-1011. doi: 10.1634/ theoncologist.2008-0131
10. Robert C, Soria JC, Spatz A, et al. Cutaneous side-effects of kinase inhibitors and blocking antibodies. Lancet Oncol. 2005; 6: 491-500. doi: 10.1016/S1470-2045(05)70243-6
11. Lipworth AD, Robert C, Zhu AX. Hand-foot syndrome (hand-foot skin reaction, palmar-plantar erythrodysesthesia): focus on sorafenib and sunitinib. Oncology. 2009; 77: 257-271. doi: 10.1159/000258880
12. Yang CH, LinWC, Chuang CK, et al. Hand-foot skin reaction in patients treated with sorafenib: a clinicopathological study of cutaneous manifestations due to multitargeted kinase inhibitor therapy. Br J Dermatol. 2008; 158: 592-596. doi: 10.1111/j.1365- 2133.2007.08357.x
13. Pragasam V, Verma R, Vasudevan B. Sorafenib and sunitinib: A dermatologist’s perspective. Ind Dermatol Online J. 2014; 5: 1-3. doi: 10.4103/2229-5178.126017
14. Pascaline BR, Céline N, Golmard JL, Audrey TS, Olivier M, Fabrice T. Early Sorafenib-induced toxicity is associated with drug exposure and UGTIA9 genetic polymorphism in patients with solid tumors: A Preliminary Study. PLoS ONE. 2012; 7(8): e42875. doi: 10.1371/journal.pone.0042875