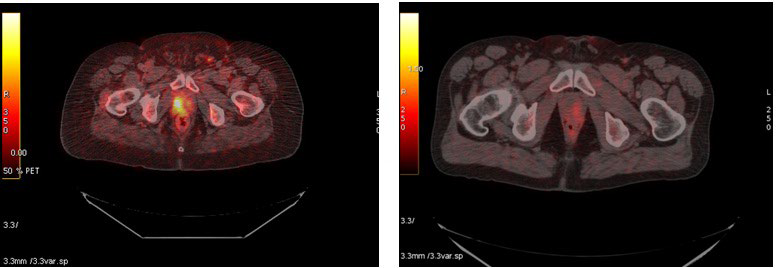
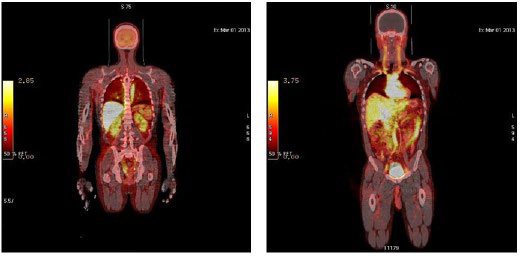
1. Blasberg RG. Imaging update: new windows, new views. Clin Cancer Res. 2007; 13(12): 3444-3448. doi: 10.1158/1078-0432. CCR-07-0936
2. Herscovitch P, Mintun MA, Raichle ME. Brain oxygen utilization measured with oxygen-15 radiotracers and positron emission tomography: generation of metabolic images. J Nucl Med. 1985; 26: 416-417.
3. Stocklin G. Tracers for metabolic imaging of brain and heart— radiochemistry and radiopharmacology. Eur J Nucl Med. 1992; 19(7): 527-551.doi: 10.1007/BF00185860
4. Agool A, SR, Kluin PM, de Wolf JT, Dierckx RA, Vellenga E. F-18 FLT PET: a non-invasive diagnostic tool for visulaization of the bone marrow compartment in patients with aplastic anemia: a pilot study. Clin Nucl Med. 2011; 36(4): 286-289. doi: 10.1097/RLU.0b013e31820aa1a1
5. Katzenellenbogen JA, Welch MJ, Dehdashti F. The development of estrogen and progestin radiopharmaceuticals for imaging breast cancer. Anticancer Research. 1997; 17(3B): 1573-1576.
6. Fried D, Garg S, Thomas A, et al. Monitoring treatment response inhigh risk prostate cancer patients using C-11 choline PET/CT. J Nucl Medicine Mol Imaging. 2010; 51: 51P.
7. Li X, Liu Q, Wang, et al. C-11 choline PET/CT imaging for differentiating malignant from benign prostate lesions. Clin Nucl Med. 2008; 33(10): 671-676. doi: 10.1097/ RLU.0b013e318184b3a0
8. Rottenburger C, Hentschel M, Kelly T, et al. Comparison of C-11 methionine and C-11 choline for PET imaging of brain metastases: a prospective pilot study. Clin Nucl Med. 2011; 36(8): 639-642. doi: 10.1097/RLU.0b013e3182175840
9. Shinoura N, Nishijima M, Hara T, et al. Brain tumors: detection with C-11 choline PET. Radiology. 1997; 202(2): 497-503. doi: 10.1148/radiology.202.2.9015080
10. Mease RC. Radionuclide based imaging of prostate cancer. Curr Top Med Chem. 2010; 10(16): 1600-1616. doi: http://dx.doi.org/10.2174/156802610793176774
11. Reske SN. Nuclear imaging of prostate cancer: current status. Urologe A. 2007; 46(11): 1485-1499. doi: 10.1007/s00120- 007-1572-6
12. Mendichovszky I, Jackson A. Imaging hypoxia in gliomas. Br J Radiol. 2011; 84 Spec No 2: S145-S158. doi: 10.1259/ bjr/82292521
13. Padhani AR, Krohn KA, Lewis JS, Alber M. Imaging oxygenation of human tumours. Eur Radiol. 2007; 17(4): 861- 872. doi: 10.1007/s00330-006-0431-y
14. Collingridge DR, Glaser M, Osman S, et al. In vitro selectivity, in vivo biodistribution and tumour uptake of annexin V radiolabelled with a positron emitting radioisotope. Br J Cancer. 2003; 89(7): 1327-1333. doi: 10.1038/sj.bjc.6601262
15. Li X, Link JM, Stekhova S, et al. Site-specific labeling of annexin V with F-18 for apoptosis imaging. Bioconjug Chem. 2008; 19(8): 1684-1688. doi: 10.1021/bc800164d
16. Garg PK, Labaree DC, Hoyte RM, Hochberg RB. [7a18F]Fluoro-17a-methyl-5a-dihydrotestosterone: a ligand for androgen receptor-mediated imaging of prostate cancer. Nucl Med Biol. 2001; 28(1): 85-90.doi: 10.1016/s0969-8051(00)00172-4
17. Garg S, Doke A, Black KW, Garg PK. In vivo biodistribution of an androgen receptor avid PET imaging agent 7-alphafluoro-17 alpha-methyl-5-alpha-dihydrotestosterone ([(18)F] FMDHT) in rats pretreated with cetrorelix, a GnRH antagonist. Eur J Nucl Med Mol Imaging. 2008; 35(2): 379-385.doi: 10.1007/s00259-007-0610-3
18. Ponde DE, Dence CS, Schuster DP, Welch MJ. Rapid and reproducible radiosynthesis of [18F] FHBG. Nucl Med Biol. 2004; 31(1): 133-138. doi: 10.1016/S0969-8051(03)00096-9
19. Lee YL, Lee YJ, Ahn SJ, et al. Combined radionuclidechemotherapy and in vivo imaging of hepatocellular carcinoma cells after transfection of a triple-gene construct, NIS, HSV1-sr39tk, and EGFP. Cancer Lett. 2010; 290(1): 129-138. doi: 10.1016/j.canlet.2009.09.004
20. Qiao H, Surti S, Choi SR, et al. Death and proliferation time course of stem cells transplanted in the myocardium. Mol Imaging Biol. 2009; 11(6): 408-414. doi: 10.1007/s11307-009- -0222-3
21. Giovacchini G, Samanes Gajate AM, Messa C, Fazio F. Increased C-11 choline uptake in pagetic bone in a patient with coexisting skeletal metastases from prostate cancer. Clin Nucl Med. 2008; 33(11): 797-798. doi: 10.1097/ RLU.0b013e318187ee35
22. Hara T, Kosaka N, Kishi H. PET imaging of prostate cancer using carbon-11-choline. J Nucl Med. 1998; 39(6): 990-995.
23. Hara T, Kosaka N, Kondo T, et al. Imaging of brain tumor, lung cancer, esophagus cancer, colon cancer, prostate cancer, and bladder cancer with [11C]choline [Abstract]. J Nucl Med. 1997; 38: 250P.
24. Attia A, V GT, Garg PK, et al. The utility of C-11 choline PET/CT for staging advanced esophageal cancer. Int J Radiat Biol Phys. 2011; 81: S314.
25. Larson SM, Morris M, Gunther I, et al. Tumor localization of 16beta-18F-fluoro-5alpha-dihydrotestosterone versus 18F-FDG in patients with progressive, metastatic prostate cancer. J Nucl Med. 2004; 45(3): 366-373.
26. DeGrado TR, Baldwin SW, Wang S, et al. Synthesis and evaluation of (18)F-labeled choline analogs as oncologic PET tracers. J Nucl Med. 2001; 42(12): 1805-1814.
27. Garg S, Lynch AJ, Doke AK, Minton RC, Garg PK. A remote controlled system for the preparation of 7 alpha-[18F]fluoro-17 alpha-methyl 5 alpha-dihydrotestosterone ([18F]FMDHT) using microwave. Appl Radiat Isot. 2008; 66(5): 612-618. doi: 10.1016/j.apradiso.2008.01.017
28. van Dongen GA, Visser GW, Lub-de Hooge MN, de Vries EG, Perk LR. Immuno-PET: a navigator in monoclonal antibody development and applications. Oncologist. 2007; 12(12): 1379- 1389. doi: 10.1634/theoncologist.12-12-1379
29. Fakih MG, Padmanabhan A. CEA monitoring in colorectal cancer. What you should know. Oncology (Williston Park). 2006; 20(6): 579-587; discussion 588, 594, 596 passim.
30. Grote T, Siwak DR, Fritsche HA, et al. Validation of reverse phase protein array for practical screening of potential biomarkers in serum and plasma: accurate detection of CA19-9 levels in pancreatic cancer. Proteomics. 2008; 8(15): 3051-3060. doi: 10.1002/pmic.200700951
31. Wiernik PH. Serum CA125 and PSA concentrations in patients with lymphoma. Clin Adv Hematol Oncol. 2008; 6(7): 527-531.
32. Kalofonos HP, Karamouzis MV, Epenetos AA. Radioimmunoscintigraphy in patients with ovarian cancer. Acta Oncol. 2001; 40(5): 549-557.doi: 10.1080/028418601750444079
33. Keane TE, Rosner IL, Wingo MS, McLeod DG. The emergence of radioimmunoscintigraphy for prostate cancer. Rev Urol. 2006; 8 Suppl 1: S20-S28.
34. Brassell SA, Rosner IL, McLeod DG. Update on magnetic resonance imaging, ProstaScint, and novel imaging in prostate cancer. Curr Opin Urol. 2005; 15(3): 163-166.doi: 10.1097/01.mou.0000165549.94663.2d
35. Sundaresan G, Yazaki PJ, Shively JE, et al. 124I-labeled engineered anti-CEA minibodies and diabodies allow high-contrast, antigen-specific small-animal PET imaging of xenografts in athymic mice. J Nucl Med. 2003; 44(12): 1962-1969.
36. Verel I, Visser GW, van Dongen GA. The promise of immuno-PET in radioimmunotherapy. J Nucl Med. 2005; 46 Suppl 1: 164S-171S.
37. Verel I, Visser GW, Boellaard R, Stigter-van Walsum M, Snow GB, van Dongen GA. 89Zr immuno-PET: comprehensive procedures for the production of 89Zr-labeled monoclonal antibodies. J Nucl Med. 2003; 44(8): 1271-1281.
38. Verel I, Visser GW, Vosjan MJ, Finn R, Boellaard R, van Dongen GA. High-quality 124I-labelled monoclonal antibodies for use as PET scouting agents prior to 131I-radioimmunotherapy. Eur J Nucl Med Mol Imaging. 2004; 31(12): 1645-1652. doi: 10.1007/s00259-004-1632-8
39. Verel I, Visser GW, Boellaard R, et al. Quantitative 89Zr immuno-PET for in vivo scouting of 90Y-labeled monoclonal antibodies in xenograft-bearing nude mice. J Nucl Med. 2003; 44(10): 1663-1670.
40. Penuelas I, Haberkorn U, Yaghoubi S, Gambhir SS. Gene therapy imaging in patients for oncological applications. Eur J Nucl Med Mol Imaging. 2005; 32 Suppl 2: S384-S403. doi: 10.1007/s00259-005-1928-3
41. Ogawa M, Kosaka N, Choyke PL, Kobayashi H. In vivo molecular imaging of cancer with a quenching near-infrared fluorescent probe using conjugates of monoclonal antibodies and indocyanine green. Cancer Res. 2009; 69(4): 1268-1272. doi: 10.1158/0008-5472.CAN-08-3116
42. Dijkmans PA, Juffermans LJ, Musters RJ, et al. Microbubbles and ultrasound: from diagnosis to therapy. Eur J Echocardiogr. 2004; 5(4): 245-256. doi: http://dx.doi.org/10.1016/j. euje.2004.02.001
43. Lindner JR. Microbubbles in medical imaging: current applications and future directions. Nat Rev Drug Discov. 2004; 3(6): 527-532. doi: 10.1038/nrd1417
44. Lanza GM, Wallace KD, Scott MJ, et al. A novel site-targeted ultrasonic contrast agent with broad biomedical application. Circulation. 1996; 94(12): 3334-3340. doi: 10.1161/01. CIR.94.12.3334
45. Unger EC, McCreery TP, Sweitzer RH, Shen D, Wu G. In vitro studies of a new thrombus-specific ultrasound contrast agent. Am J Cardiol. 1998; 81(12A): 58G-61G. doi: 10.1016/ S0002-9149(98)00055-1
46. Niu Z, Fang Y. Surface-enhanced Raman scattering of single-walled carbon nanotubes on silver-coated and gold-coated filter paper. J Colloid Interface Sci. 2006; 303(1): 224-228. doi: 10.1016/j.jcis.2006.07.039
47. Nie S, Emory SR. Probing Single Molecules and Single Nanoparticles by Surface-Enhanced Raman Scattering. Science. 1997; 275(5303): 1102-1106. doi: 10.1126/science.275.5303.1102
48. Zavaleta CL, Smith BR, Walton I, et al. Multiplexed imaging of surface enhanced Raman scattering nanotags in living mice using noninvasive Raman spectroscopy. Proc Natl Acad Sci U S A. 2009; 106(32): 13511-13556.doi: 10.1073/pnas.0813327106