INTRODUCTION
Coronary artery ectasia (CAE) is defined as dilatation of an arterial segment to a diameter at least 1.5 times that of the adjacent normal coronary artery.1 It is found to occur in 1.2-4.7% of patients undergoing coronary angiography (CAG).2 The etiologies of CAE are atherosclerosis in 50% cases, congenital in 20-30% while 10-20% are associated with connective tissue disorders.3
CAE is not a benign entity as it leads to sluggish or turbulent coronary blood flow, with exercise induced angina and myocardial infarction (MI), regardless of the severity of coexisting stenotic coronary disease, arrhythmia and sudden cardiac death. This is due to repeated dissemination of microthrombi to segments distal to the ectasia, or to thrombotic occlusion of the dilated vessel. Slow flow during a procedure or spontaneously may also be a causative factor.3
Acute coronary syndromes such as MI in the setting of underlying CAE pose serious therapeutic challenges for the operator because 1) No clear-cut guidelines exist regarding what treatment strategy is to be used-fibrinolysis, balloon angioplasty or stenting, 2) Dedicated hardware especially large diameter coronary stents required for such cases are not available and 3) High thrombus burden poses significant challenges.
We recently encountered one such case where a patient presented with acute ST elevation MI and had severe CAE which was successfully treated using a renal stenting, there being no coronary stent more than 5 mm being manufactured.
CASE SERIES
A fifty-six-year-old hypertensive male presented to the Emergency Department with severe retrosternal discomfort of 2 hours duration. An urgent electrocardiogram (ECG) was suggestive of acute anterior wall MI. His heart rate was 72 per minute; blood pressure 130/80 mmHg, Cardiovascular and respiratory system examination were unremarkable. He received a loading dose of 180 mg ticagrelor, 325 mg as aspirin and 80 mg of atorvastatin. After an informed consent was obtained a coronary angiogram was immediately performed which revealed severe CEA. The right coronary artery (RCA) and left circumflex (LCx) were ectatic but had no obstructive lesions; however the left anterior descending artery apart from being ectatic (approximately 7 mm in diameter) was 100% occluded proximally (Figure 1A and 1B). The technical challenges posed by these angiographic findings were discussed with the patient and his relatives and the options of either fibrinolytic therapy or percutaneous coronary intervention (PCI) and implantation of a peripheral stent in the left anterior descending artery (LAD) were offered. They opted for the latter treatment option after all pros and cons were explained. The left coronary ostium was engaged using a Judkins left (JL 4) 7F guiding catheter. The lesion was crossed using an All star 0.014”×180 cms percutaneous transluminal coronary angioplasty (PTCA) wire (Abbot Vascular, Santa Clara, CA, USA) and significant thrombus burden was noted in the LAD (Figure 2A). Injection Abciximab bolus and infusion was initiated as per protocol. Repeated thrombus aspiration was carried out using a Thrombuster II catheter (Kaneka Corporation, Osaka, Japan) and thrombotic material was retrieved. Subsequently once reasonably satisfactory flow was achieved in the LAD and the culprit lesion identified (Figure 2B) it was stented using a 7×18 mm RX Herculink Elite Renal Stent (Abbot Vascular, Santa Clara, CA, USA) deployed at 11 atmospheres (Figure 2C). The stent system has a shaft length of 135 cms. Thrombolysis in Myocardial Infarction (TIMI) III flow was achieved in the vessel (Figure 2D) with resolution of symptoms. Post PCI period was uneventful and patient was discharged successfully on dual antiplatelet therapy (Aspirin and Ticagrelor), statins, beta blocker and Angiotensin converting enzyme inhibitor. The patient is doing well 4 weeks after the procedure.
Figure 1A: Right anterior oblique (RAO 30º) shows an ectatic right coronary artery.
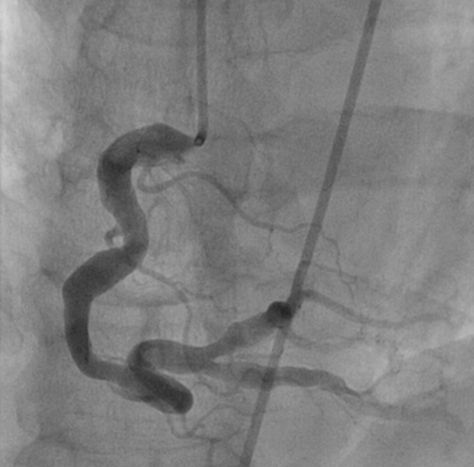
Figure 1B: Left anterior oblique (LAO) caudal or spider view shows ectatic left sided coronaries and a 100% occluded LAD artery.
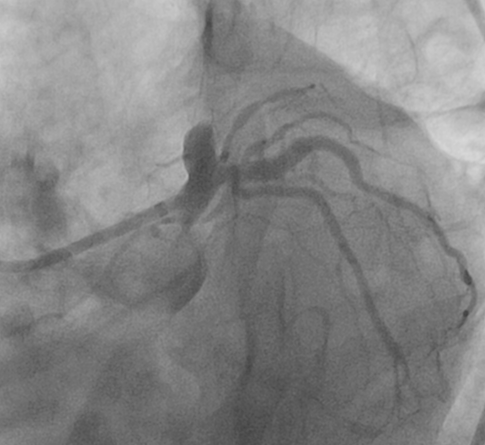
Figure 2A: Caudal view after the wire crossed the lesion in LAD shows a heavy thrombus burden.
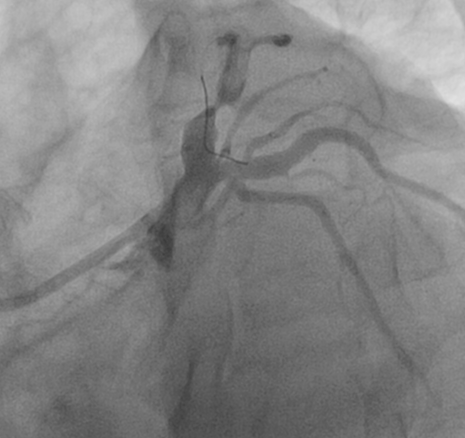
Figure 2B: RAO caudal view shows the target lesion in LAD.
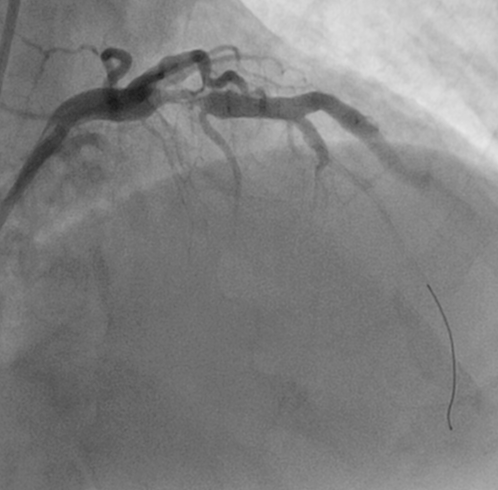
Figure 2C: Stent being placed across the lesion in the LAO caudal view.
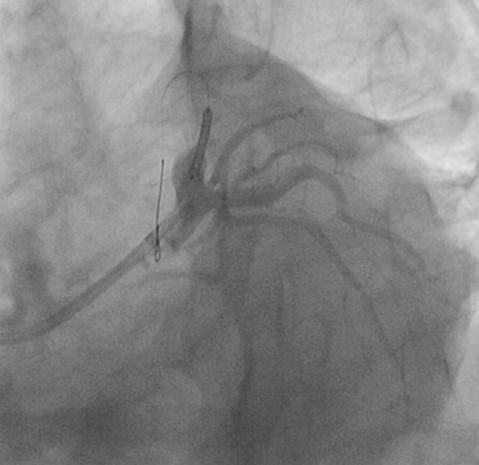
Figure 2D: LAO caudal view after the stent is deployed.
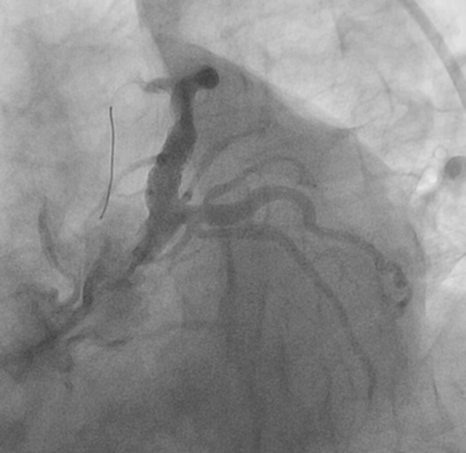
DISCUSSION
CAE poses significant management challenges. Various medical treatments tried include empiric chronic anticoagulation,4 dual antiplatelet therapy and medications with vasodilatory properties (nitrates, calcium channel blockers, angiotensin-converting-enzyme inhibitor (ACE) inhibitors and trimetazidine). However no prospective studies have been carried out and these cannot be recommended at present as a standard of care. Nitrates theoretically seem to be appropriate in patients with CAE due to their ability to cause coronary dilatation, however on the contrary they have been shown to exacerbate myocardial ischemia.5 Stent implantation is recommended only when significant stenosis accompanies the CAE and medical therapy has not been successful. This is because these lesions have heterogeneous morphologies, large caliber making it difficult to determine stent type, size and complete expansion. If the target lesion is not suitable for stent implantation, surgical excision or ligation with a bypass graft could be choice of treatment.3
However a real dilemma is when one is faced with a CAE in an acute MI situation as in the present patient. The treatment options include fibrinolysis, plain balloon angioplasty or stenting with a peripheral stent. Oh et al1 described two cases of ACS in CAE setting. One patient underwent plain balloon angioplasty while the other received a 6×24 mm Genesis peripheral stent. Agarbasli et al6 reported 3 cases of coronary angioplasty with intra- therapeutic peripheral biliary stents for CAE. This was the first report of using a peripheral biliary stent for coronary revascularization. Rha et al reported the use of two parallel stents (3.5×18 mm sirolimus eluting stents) in a patient with CAE.7 In the present case the vessel diameter was 7 mm and the renal stent available on shelf had the desired dimensions and also a shaft length of 135 cms such that it could be delivered to the desired site in the coronary tree. As far as options of Antiplatelet therapy are concerned usually after placing peripheral stents dual antiplatelet therapy (DAPT) is continued for a month. Beyond the first month a single Antiplatelet agent usually aspirin is continued indefinitely. In this case as a peripheral stent was used in coronary location we plan to continue DAPT for 1 year and then switch over to single Antiplatelet drug in accordance with ACC/AHA guidelines for coronary stents. To the best of our knowledge this is the first case in literature where a renal stent has been used in the coronary tree.
CONCLUSION
Surprises may spring up at any given time in the cardiac catheterization laboratory. Unusual scenarios such the one mentioned above may call for quick and out of the box thinking. Peripheral stents may help revascularize ectatic vessels in which routinely available coronary stents cannot be used due to sizing constraints.
Industry in collaboration with interventional cardiologists needs to develop hardware for PCI of vessels with ectasia. In view of the mismatches in the size of these vessels a self expanding stent seems the most appropriate.
CONFLICTS OF INTEREST
The authors declare that they have no conflicts of interest.











