INTRODUCTION
Trigeminal neuropathy is described as the most common neurological cause of dropped jaw in dogs.1 Also referred to as idiopathic trigeminal neuropathy (ITN), trigeminal neuritis, and trigeminal neurapraxia, this condition primarily affects the motor branches of the trigeminal nerves bilaterally, although associated loss of facial sensation and Horner’s syndrome are described.2 The condition is self-limiting and is characterised by spontaneous full recovery. The published literature on magnetic resonance imaging (MRI) findings for this condition is limited to two cases, and describes non-specific changes within the trigeminal nerve.3 To the author’s knowledge, this is the first reported case of suspected ITN with MR signal changes within the brain.
CASE HISTORY
An 8-year 9-month old male Staffordshire bull terrier presented to the neurology department of Dick White Referrals (Newmarket, UK) with a 4-day history of an inability to close the jaw, excessive salivation and difficulty eating and drinking. On neurological examination, there was a complete absence of jaw tone with normal visual and palpable temporal muscle mass. Facial sensation was normal, as was the gag reflex, and no evidence of Horner’s syndrome was seen. Remaining cranial nerve tests including menace response, pupillary light reflex, dazzle reflex, corneal and palpebral reflexes and oculovestibular response tests were normal. Neuroanatomical localisation was to bilateral trigeminal nerve (motor branch) dysfunction. A complete blood count and routine biochemistry panel were unremarkable. MRI was undertaken to further pursue a diagnosis.
Magnetic resonance (MR) images were acquired with a 0.4T magnet (Hitachi Aperto, Japan). Imaging sequences include SAG FSE T2, TRANS FSE T2, TRANS T2 Flair, TRANS T2* GRE, TRANS SE T1 and post contrast SE T1 in 3 orthogonal planes. Diffusion weighted images were also acquired in the transverse plane.
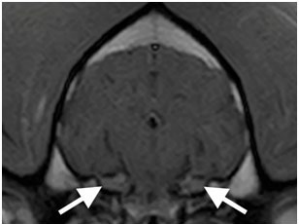
On the T2 weighted images symmetrical, ill-defined hyperintensities within the pons at the level of the trigeminal nuclei were seen (Figure 1). These areas appeared isointense on T1W images. There was mild heterogeneous contrast enhancement of the trigeminal nerve and ganglion bilaterally (Figure 2), with no enhancement seen within the pons itself. No mass lesion or mass effect was seen with the remainder of the brain parenchyma unremarkable on all sequences. There was no evidence of muscle atrophy or signal changes within the masticatory muscles. Regional lymph nodes appeared normal.
Figure 1: Transverse T2 Weighted FLAIR Image at the Level of the Pons. Bilateral Hyperintensities (White Arrows) are seen in the Regions of the Trigeminal Nuclei.
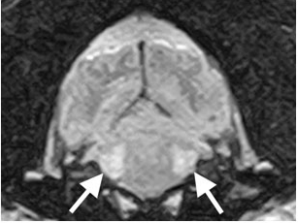
Figure 2: Transverse T1 Weighted Post-Contrast Image of the Brain Showing Normal Appearance and Enhancement of the Trigeminal Nerves at the Level of the Ganglia (White Arrows).
RT-PCR on cerebrospinal fluid (CSF) for Distemper, Toxoplasma, Neospora, Bornavirus, Borrelia, Ehrlichia, Leishmania, Canine Herpes, Parvovirus and Minute virus were negative. Given the bilaterally symmetrical changes and in absence of mass effect neoplasia was considered less likely and a presumptive diagnosis of inflammatory or idiopathic trigeminal neuropathy was made. A CSF sample obtained from the cistern magnum after MR imaging showed albuminolocytologic dissociation, with protein 0.41 g/L (ref: <0.30).
The dog was hospitalised on fluid maintenance and supportive feeding. Treatment was started with 0.2 mg/kg dexamethasone (Dexadreson; MSD Animal Health) intravenously and 4 mg/kg gabapentin (Gabapentin; Zentiva) per os every 24 hours, 2 mg/kg ranitidine (Zantac; GlaxoSmithKline) every 12 hours, and 90 mg/m2 lomustine (Lomustine; medac). The dog began to persistently regurgitate for the 1st 24 hours in hospital. A single left lateral thoracic radiograph was unremarkable with no evidence of megaoesophagus. The regurgitation fully resolved on 1 mg/kg maropitant (Cerenia; Zoetis) every 24 hours and 1 mg/kg/24 hours metoclopramide (Emeprid; Ceva) continuous rate infusion.
Over 6 days of hospitalisation, the gabapentin dose was increased to 4 mg/kg every eight hours. Marginal improvement in the jaw tone was noted, and the dog was coping well with assisted feeding, drinking and per os medication. He was discharged to continue with 0.08 mg/kg dexamethasone every 24 hours and 4 mg/kg gabapentin every eight hours, with a 14 days course of 2 mg/kg ranitidine every 12 hours and 0.4 mg/kg metoclopramide every 12 hours.
At follow-up with the owner 2 weeks later by phone call he was continuing to do well, with gradually returning function and strength of the jaw. The owners reported he still required assistance to eat and drink but was improving with significantly reduced hypersalivation. The owners had noticed significant atrophy of the masseter muscles bilaterally, however, he had only recently returned to chewing food. At this point, the dexamethasone was reduced to 0.04 mg/kg every 24 hours and all other medications stopped. At a further phone call 2 months after discharge, the owners reported that gradual improvement had continued and he had recently become clinically normal in their opinion. All medications had finished 2 weeks prior to this check-up, and no further check-ups were advised unless signs of recurrence were seen. A final phone update 8 months after discharge confirmed no deterioration with normal jaw function.
DISCUSSION
Flaccid mandibular paralysis or ‘dropped jaw’ is caused by dysfunction in the motor branch of the trigeminal nerve, with potential aetiologies in dogs including infectious, inflammatory, neoplastic and idiopathic.1 ITN is primarily a dysfunction of the motor tracts with associated ‘dropped jaw’.2 A minority of cases have facial sensory deficits and/or unilateral Horner’s syndrome, although absence of concurrent neurological signs is common.4 When cases of ITN were reviewed, the only tests to show abnormal results in most cases were CSF analysis and electromyography (EMG).2 On CSF analysis, mild to moderate elevations in total protein, and mild elevations in nucleated cell counts, were the most common findings, consistent with the mild elevation in protein levels seen in this case.
A suspected diagnosis of trigeminal neuritis is confirmed when the clinical signs self resolve, usually within 2-3 weeks, although this has been reported to range from 4 to 63 days.1,4 Interestingly, the use of steroid therapy has showed no significant difference in resolution time.2 Given this pattern of spontaneous full clinical recovery, there is no definitive histological evidence of the disease from post-mortem, and therefore a poor understanding of its aetiology, although an inflammatory cause is suspected.
Reported findings on MR imaging of ITN are very limited. Given the resolving nature of the clinical signs, MR is often not undertaken when a diagnosis of ITN is suspected, it’s main use has been to rule out neoplasia in cases of unilateral dysfunction, or where clinical signs progress or fail to resolve.5,6 In two dogs with histological confirmation of neuritis, both had diffuse enlargement of the trigeminal nerves described within the calvarium and trigeminal canal, but with no discrete mass lesion or mass effect seen.3 The nerves were isointense on T1W precontrast and PDW images in both dogs, with the nerves hyperintense on T2W images in one case and isointense on T2W images in the other. Either heterogeneous or homogeneous enhancement of the nerve was seen on post-contrast T1W images. Post-contrast enhancement of the ganglia was seen in this case however, enhancement of the trigeminal ganglia with or without enhancement of the associated nerves has been shown to be a normal finding.6 In cases of nerve-sheath neoplasia, unilateral discrete contrasting enhancing mass lesions are described, with or without displacement of the adjacent brainstem.3,7,8 No cases of idiopathic trigeminal neuropathy with MR changes within the brainstem, were found on review of the literature.
CONCLUSION
Giving the lack of histological confirmation for diagnosis, a broad treatment protocol was used. The dog progressed to full clinical improvement and remained so on withdrawal of all medications. This clinical behaviour is not consistent with the other considered differentials, and therefore this case is presented as a likely severe case of idiopathic trigeminal neuropathy with novel intra-cranial findings on MR.
CONFLICTS OF INTEREST
The authors declare they have no conflicts of interest.
CONSENT
The owner has given permission for publication of any material related to this case.







