INTRODUCTION
Inflammatory Breast Cancer (IBC) is one of the most aggressive forms of breast cancer. IBC is a rare form of breast cancer which is diagnosed according to clinical presentations, including rapid onset of erythema and edema of the skin and breast (peau d’orange). Patients with locally advanced IBC usually have a poor prognosis, with five year survival rates around 30-50%.1,2 Despite multi-modality treatments, local recurrence is common. Significantly higher incidence of loco-regional recurrences are found in IBC patients compared to those with noninflammatory breast cancers.2
Local recurrence has been linked to the presence of radioresistant Breast Cancer Stemlike Cells (BCSC). These cells, bearing the phenotype of CD44high/CD24low/-, resemble normal mammary stem cells in that they are capable of undergoing continuous self-renewal. BCSC have been shown to be resistant to radiation through several mechanisms including over expression of DNA repair enzymes, cell cycle checkpoint proteins and survival proteins.3,5,6 Due to their intrinsic radioresistance and self-renewal properties, BCSC are thought promote to tumor regrowth after radiation.
Soy isoflavones, such as genistein, have been reported to have anti-tumor properties in multiple tissue types. Treatment of non-inflammatory breast cancers with genistein has been shown to decrease proliferation and sensitize breast cancer cells to chemotherapy.7 Furthermore, genistein has been shown to decrease BCSC populations in non-inflammatory breast cancer.8 While genistein has been reported to have a radioprotective effect on normal tissues,9 treatment of non-inflammatory breast cancer cells with high doses (µM range) of genistein has been shown to increase radiation induced death through modulation of cell cycle and reduced expression of DNA repair proteins.10 Although treatment of cancer cells with high doses of genistein may have a growth inhibitory effect, dietary intake may result in long term exposure to much lower doses. The effect of chronic exposure to low doses of genistein is unclear. The aim of this study is to determine the effects of long term exposure of inflammatory breast cancer cell lines to low doses of genistein compared to short term high dose treatment.
METHODS
Cell Lines
Cell lines were purchased from Asterand Inc. (Detroit, MI, USA). SUM149 cells were cultured in Ham’s F12 medium supplemented with 5% (v/v) FBS, 1% (v/v) Antiobiotic Antimycotic, 1% (v/v) Insulin-Transferrin-Selenium (ITS) and 1 μg/mL hydrocortisone (Sigma). IBC3 cells were cultured in Ham’s F12 medium supplemented with 10% (v/v) FBS, 1% (v.v) Antibiotic Antimycotic, 100 ng/mL hydrocortisone and 2 μg/mL insulin. Genistein was purchased from Sigma and was resuspended in DMSO at a stock concentration of 20 mM. All media reagents were purchased from Life Technologies unless otherwise noted.
Cell Survival Assay
Cells were plated at a density of 7000 cells per well in 96 well plates and cultured overnight. Cells were treated with genistein at the indicated concentrations and assayed for survival 72 hours later using the MTT Cell viability kit assay (Biotium Inc, Hayward, CA, USA). Data is represented as percent of untreated cells.
Mammosphere Forming Assay
Cells were removed by trypsinization and washed with culture media, followed by 3 washes with PBS. Cells were resuspended in MEM supplemented with 1X B27 (Invitrogen), 20 ng/ mL Epidermal Growth Factor (EGF; Invitrogen), 20 ng/mL basic Fibroblast Growth Factor (bFGF; Invitrogen). For treatment groups, genistein was added at the indicated concentration. For long-term exposure, cells were cultured with 40 nM genistein for 2-3 weeks prior to plating for the mammosphere assay. Cells were seeded into ultra-low attachment plates (Corning Life Sciences, Salt Lake City, UT, USA) at a density of 1000-2000 cells per well. Cells were grown for 10-14 days and spheres were counted. Only spheres that were at least 50 microns or greater in size were counted. Vehicle control experiments were carried out with a dilution of DMSO only in cell culture media. Data is represented as % control spheres.
Flow Cytometry
For long-term treatments, cells were treated with 40 nM genistein in normal culture media for 3 weeks prior to the experiment. For short term treatments, cells were treated with 10 µM genistein 48 hours prior to analysis. Cells were stained with PE-conjugated CD44 and FITC-conjugated CD24 (Abcam, Cambridge, MA, USA) for 30 min at RT. Samples were centrifuged, washed, and passed through BD Falcon cell strainer cap tubes for FACS analysis. Samples were analyzed on a BD FACSAria II flow cytometer and samples were gated on an unstained control.
Clonogenic Assay
Cells were treated with 40 nM genistein for 3 weeks prior to the assay or with 10 µM genistein at the time of plating. Cells were seeded into 6 well plates at densities ranging from 50-5000 cells per well, in triplicate. Plates were irradiated at the indicated dose using an X-ray microirradiator, X-RAD225 (Precision X-ray, North Branford, CT, USA) and cultured for 10-14 days. Colonies were fixed with 10% formalin solution and stained with a 0.05% solution of crystal violet. Vehicle control experiments were carried out with a dilution of DMSO only. Individual colonies were counted and colony forming efficiency was determined as colonies counted/cells plated.
Statistical Analysis
Statistical analysis was performed using Graph Pad Prism 6 software (Graph Pad, La Jolla, CA, USA). The means values of data were evaluated using ANOVA followed by an unpaired test. For all tests, P values less than 0.05 were considered to be significant.
RESULTS
Treatment of IBC Cell Lines with High Dose Genistein decreases Cell Viability
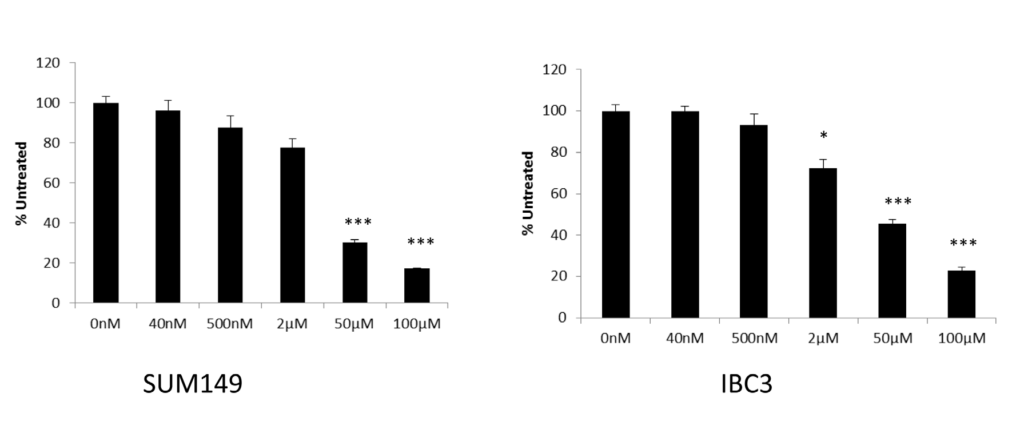
To determine the effects of high (µM) and low dose (nM) genistein on survival of inflammatory breast cancer cells we treated two cell lines, SUM149 and IBC3, with genistein and measured cell viability using an MTT-based assay 72 hours after treatment. Decreased cell survival was observed in both lines after treatment with high dose genistein (Figure 1) with a LD50 around 50 µM in the IBC3 line. SUM149 cells were slightly more sensitive, with only 30% survival at 50 µM compared to
controls. No significant effect on cell viability was observed for cells treated with doses lower than 500 nM in either short term or long term (>2 weeks). Although not significant, a slight decrease in cell doubling times was observed in cells treated with low dose genistein for greater than 2 weeks (data not shown).
Figure 1: Treatment of IBC cell lines with high dose genistein decreases cell growth and survival. MTT assay of SUM149 and IBC3 cells treated with genistein at the indicated concentrations. Cell viability was measured 72 hours after treatment. Data is reported as percent of untreated cells. Error bars represent standard deviation. Asterisk represent significance relative to untreated cells, *p <0.5, ***p <.005.
 Long Term Culture of IBC Cells with Low Dose Genistein decreases Cancer Stem Cell Populations
Long Term Culture of IBC Cells with Low Dose Genistein decreases Cancer Stem Cell Populations
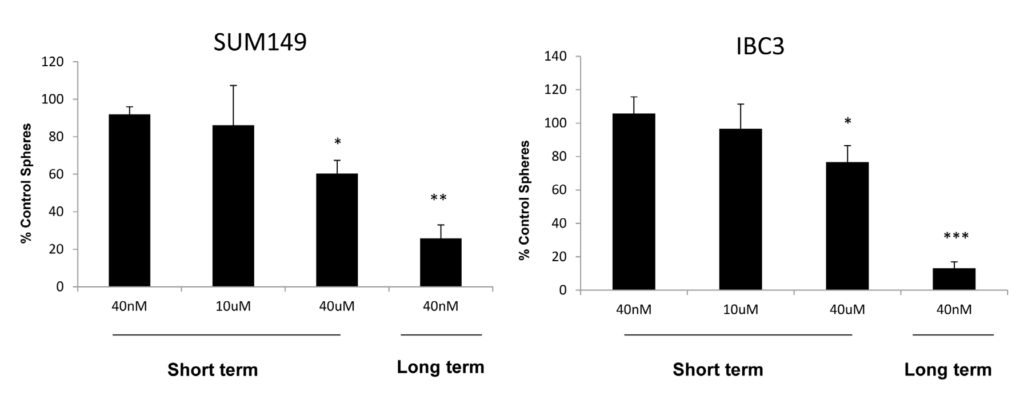
The ability to form mammospheres in three dimensional cultures has been associated with self- renewal and tumor initiating properties. We sought to determine the effect of high and low dose genistein on mammosphere forming ability of IBC cell lines. Cells were treated either short term or long term with genistein or vehicle carrier and plated in mammosphere forming assays. As shown in Figure 2, treatment of cells with low dose genistein at the time of plating had no effect on mammosphere formation in both lines. A significant decrease in mammosphere forming ability was observed upon treatment with high doses of genistein at the time of plating. However, the greatest decrease in mammosphere formation was observed in cells which were cultured long term with low doses of genistein, suggesting a decrease in self-renewal of these cells.
Figure 2: Long term treatment with low dose genistein reduces mammosphere formation. SUM149 and IBC3 cells were cultured with 40 nM genistein for 3 weeks and plated in mammosphere assays. Alternatively, micromolar concentrations of genistein were added at the time of plating. Cells were grown for 10-14 days and spheres were counted. Data is expressed as % of spheres counted compared to number of spheres counted in control cells that received treatment with the vehicle control only. A significant decrease in sphere formation was observed in cells treated with long term low dose genistein. Error bars represent standard deviations. Asterisk represent significance relative to untreated cells, *p<.05, ***p<.005.
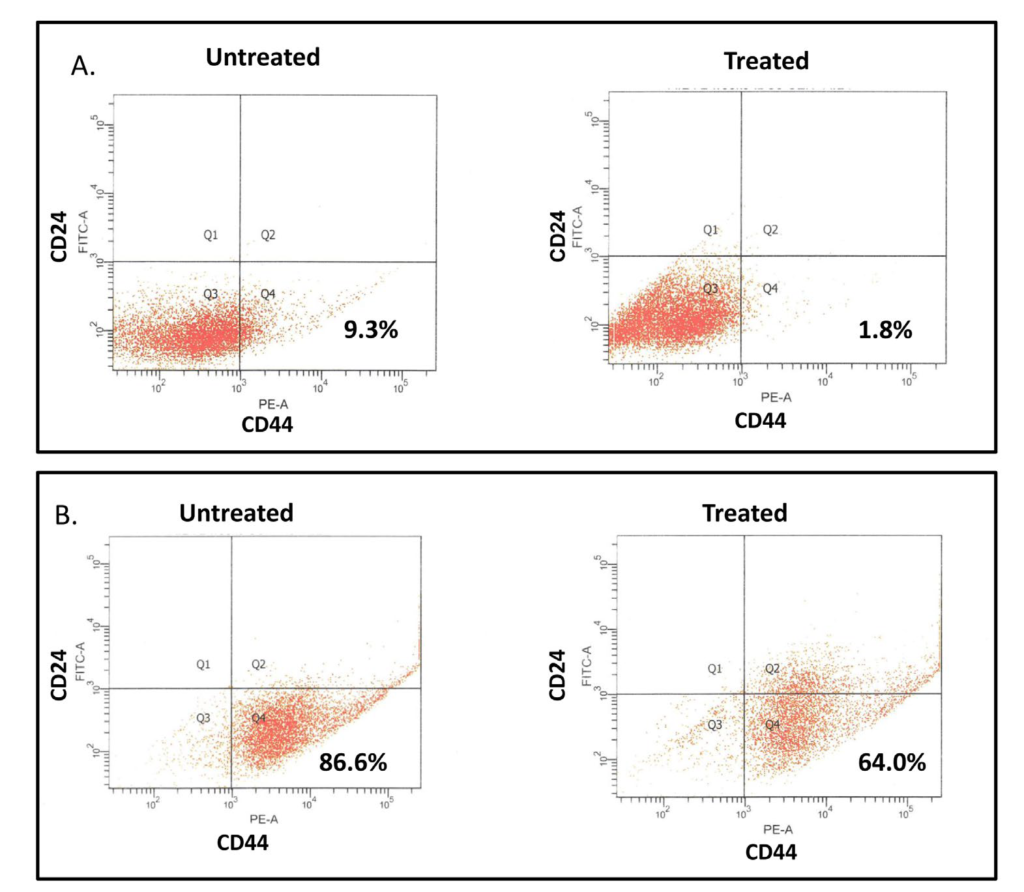
 To further confirm the effects of long term exposure of IBC cells to low dose genistein on cancer stem cell populations in IBC cell lines, we examined the CD44high/CD24low/- populations in cells cultured with and without 40 nM genistein for three weeks. A significant reduction in CD44high/CD24low/- population was observed in treated cells in both lines (Figure 3). No significant changes were observed in cells treated 48-72 hours prior to analysis, even with higher doses (data not shown). This finding indicates that long term treatment with lower doses of genistein may have beneficial effects on tumor cells through a decrease in cancer stem cells.
To further confirm the effects of long term exposure of IBC cells to low dose genistein on cancer stem cell populations in IBC cell lines, we examined the CD44high/CD24low/- populations in cells cultured with and without 40 nM genistein for three weeks. A significant reduction in CD44high/CD24low/- population was observed in treated cells in both lines (Figure 3). No significant changes were observed in cells treated 48-72 hours prior to analysis, even with higher doses (data not shown). This finding indicates that long term treatment with lower doses of genistein may have beneficial effects on tumor cells through a decrease in cancer stem cells.
Figure 3: Long term treatment with low dose genistein reduces stem cell populations. IBC3 (A) and SUM149 (B) cells were cultured with and without 40 nM genistein for three weeks and analyzed for CD44 and CD24 expression by flow cytometry. A significant decrease in the stem cell population characterized by high CD44 and low CD24 expression was observed in treated cells.
Long Term Culture of IBC Cells with Low Dose Genistein increases Radiosensitivity
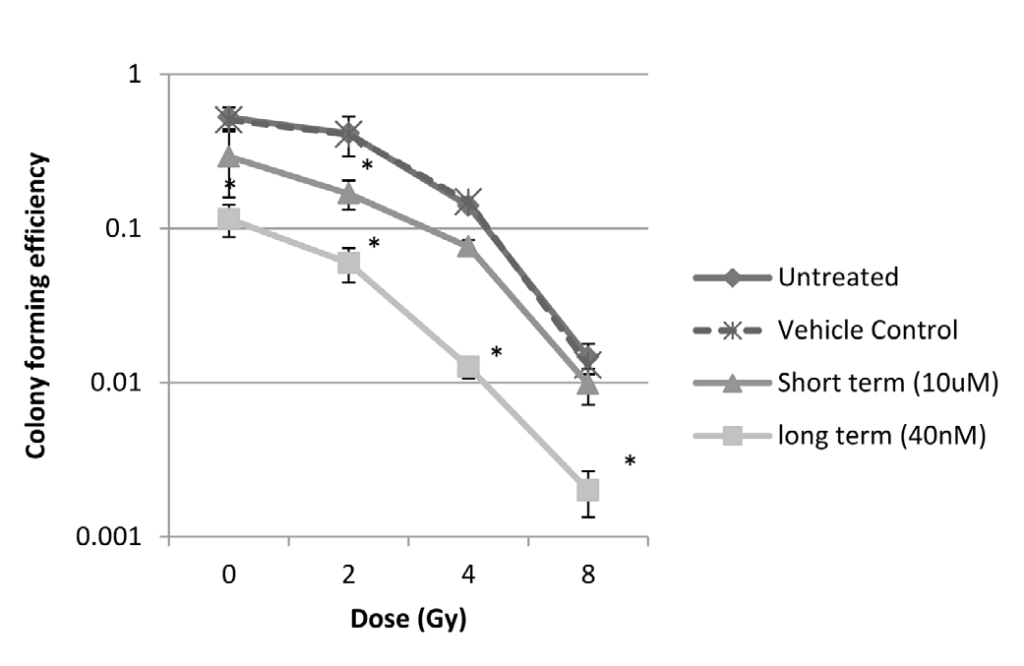
Previous studies have shown that breast cancer stem cell populations have increased resistance to radiation induced cell death. We therefore wanted to examine the effect of low and high dose genistein treatment of IBC cells after exposure to ionizing radiation. SUM149 cells were cultured with 40 nM genistein for 3 weeks (long term) or were treated 24 hours prior to radiation exposure with 10 µM genistein or vehicle carriers. Cells were irradiated at the indicated doses and colony formation was analyzed compared to non-irradiated controls. No differences were observed between untreated cells and cells treated with the vehicle carrier. Treatment with both 10 µM or long term treatment with 40 nM genistein decreased colony formation in non-irradiated cells (Figure 4). Combination treatment with 10 µM and ionizing radiation increased radiosensitivity at low doses of radiation, but had minimal effect on colony formation at higher doses compared to cells that were treated with radiation only. The radiosensitization effect was more significant in cells that were cultured long term with low dose genistein, even at relatively high radiation doses. This finding is consistent with the previous experiment which showed that long term culture of IBC cell lines with low dose genistein decreases stem-like populations, and it indicates that genistein may sensitize tumors to radiation through a decrease in resistant cancer stem cells.
Figure 4: Clonogenic assay. Genistein sensitizes IBC cell lines to radiation. SUM149 cells were cultured with and without 40 nM genistein for three weeks and exposed to ionizing radiation. Alternatively, cells were pretreated with micromolar doses of genistein or vehicle control for 24 hours prior to radiation. Cells were plated for clonogenic assay and grown for 10 days, at which time colonies were counted. Data is expressed as colony forming efficacy (log[colonies counted/cells plated]). Error bars represent standard deviations. Asterisk represent significance p <.001, relative to vehicle control cells.
 DISCUSSION
DISCUSSION
Local recurrence after treatment with radiation is a common event in IBC, and is associated with poor survival. Recurrence is thought to be due to the presence of radioresistant cancer stem cells. Here we show that long term treatment with low doses of genistein may help to decrease growth of cancer stem cells, and increase radiation induced cell death. Although treatment of IBC cell lines with higher doses of genistein induced cell death, minimal effect was observed on cancer stem cell populations. These findings suggest that long-term exposure may decrease proliferation of stem like populations and lead to reduced levels of resistant cells. Previous studies have shown micromolar doses of genistein increase radiation induced cell death in non-inflammatory breast cancer cell lines by induced growth arrest and accumulation of cells in the most radiosensitive phase of the cell cycle.10 Our data suggest that high dose genistein may act on inflammatory breast cancer cells in a similar matter, as growth arrest was detected after treatment with micromolar doses. However, this mechanism does not explain the radiosensitizing effect of long term low dose genistein as this had a minimal effect on growth of the total population of cancer cells. We propose that cancer stem cells may be more sensitive to genistein-induced growth arrest. Therefore, long-term treatment with genistein may decrease this population of radioresistant cells overtime, leading to a significant decrease in colony formation after radiation.
Genistein is a phytoestrogen that is found in soy and other legumes. Although the role of dietary soy in breast cancer has not been clear, a meta-analysis has shown that consumption of dietary soy after breast cancer diagnosis is associated with lower rates of recurrence and better survival.11 The components of dietary soy that are responsible for the improved prognosis have not been clearly identified. Soy food contains many components such as proteins, peptides, phenolic acids, saponins and isoflavones which may provide anti-cancer benefits.12 In particular, soy isoflavones such as genistein and daidzein have been directly implicated in decreases of tumor growth. While the majority of studies looking at anti-cancer properties of genistein tested concentrations at the micromolar level, our study shows that long term exposure to very low dose genistein may have an effect as well. Soy isoflavones are subjected to extensive metabolism and disposition after human digestion. Phase II metabolism by glucuronidation is a major pathway for the isoflavones, and soy isoflavones circulate in several molecular forms, including glucuronide and sulfate conjugates, freely circulating aglycons, and protein-bound aglycons.13 While a majority of absorbed isoflavones are excreted as conjugates into the urine, a smaller percentage undergoes enterohepatic recycling.14As a result, normal physiological genistein concentration (total genistein including that from conjugated genistein after enzymatic hydrolysis) were measured as a range of 0-4192 nM with a mean of 501.9 nM from the serum of Japanese women (n=125), and ranged from 0-4092 nM with a mean of 492.7 nM in the serum of men (n=102) over forty years of age.15 In a recent report, the plasma concentrations of unconjugated genistein (free aglycon) in 20 healthy adults (10 women and 10 men) after consumption of soy food (soy milk, soy nuts, or Tempeh) were extremely low and no differences were observed between men and women.16 When all the data in that study were pooled, the mean unconjugated genistein concentration was extremely low at 0.46±0.12 ng/mL.16 Actual cell uptake of soy isoflavones due to dietary consumption is thus very low and might be within nanomolar level. In this study, we observed differences between long term treatments of nanomolar doses of genistein compared to acute micromolar doses of genistein. However, further study needs to be done to determine how these doses mimic long term consumption of dietary soy. In summary, we show that treatment of inflammatory breast cancer with micromolar concentrations of genistein results in growth arrest, decreased survival, and partial radiosensitization at lower radiation doses. Long term exposure to nanomolar concentrations had minimal effect on cell growth, but it decreased stem cell populations and led to a significant decrease in colony formation following radiation treatment. Thus, our study suggests a role for long-term low dose genistein in the prevention of local recurrence, through a decrease in cancer stem cell populations. Further investigation is necessary to determine the in vivo effect of low dose genistein on stem cell populations in inflammatory breast cancer.
FUNDING ACKNOWLEDGEMENTS
This project was supported by the Delaware INBRE program, with a grant from the National Institute of General Medical Sciences – NIGMS (8 P20 GM103446-13) from the National Institutes of Health.
CONFLICTS OF INTEREST:
None.

 Long Term Culture of IBC Cells with Low Dose Genistein decreases Cancer Stem Cell Populations
Long Term Culture of IBC Cells with Low Dose Genistein decreases Cancer Stem Cell Populations To further confirm the effects of long term exposure of IBC cells to low dose genistein on cancer stem cell populations in IBC cell lines, we examined the CD44high/CD24low/- populations in cells cultured with and without 40 nM genistein for three weeks. A significant reduction in CD44high/CD24low/- population was observed in treated cells in both lines (Figure 3). No significant changes were observed in cells treated 48-72 hours prior to analysis, even with higher doses (data not shown). This finding indicates that long term treatment with lower doses of genistein may have beneficial effects on tumor cells through a decrease in cancer stem cells.
To further confirm the effects of long term exposure of IBC cells to low dose genistein on cancer stem cell populations in IBC cell lines, we examined the CD44high/CD24low/- populations in cells cultured with and without 40 nM genistein for three weeks. A significant reduction in CD44high/CD24low/- population was observed in treated cells in both lines (Figure 3). No significant changes were observed in cells treated 48-72 hours prior to analysis, even with higher doses (data not shown). This finding indicates that long term treatment with lower doses of genistein may have beneficial effects on tumor cells through a decrease in cancer stem cells.
 DISCUSSION
DISCUSSION



