INTRODUCTION
The interaction of xenobiotics with the biological system is a multifaceted phenomenon, which comprises interplay between the environment, the host and the chemical substance. Chemical contagion as a result of pesticide introduction has been assumed as one of the factors for the deterioration of natural fauna. Indiscriminate application of pesticides is usually accompanied with serious problems of pollution and health hazards.1
Pyrethroids, derivatives of natural pyrethrins have been well known for their high effectiveness against insects and can be easily biodegraded than other types of pesticides.2 In spite of claims of low mammalian toxicity of pyrethroid, several investigations reported the toxicological evidence of pyrethroid among various species of animals.3 Pyrethroids are easily absorbed through gastrointestinal and respiratory tract due to their lipophilic nature and also make them easier to be stored in the lipid rich internal tissues like body fat, skin, liver, kidney, central and peripheral nervous systems. Hemato-biochemical studies are important for the analysis of the functional status of animals to suspected toxic agents. It may act as a strong evidence against toxicity of contaminated pyrethroid insecticides. Recent reports have clarified that exposure to pyrethroid leads to a significant modifications in hematological findings.4 On the other hand the liver is the first organ encountered when toxicants enter into the body. The liver was found to accumulate a huge pyrethroid residues as it is the primary site for pyrethroid metabolism. A large number of man-made chemicals such as pesticides stated to produce liver damage.5 Several investigations also reported that pesticides adversely affect the testicular functions in experimental animals6-9 as well as they are potent endocrine disrupters.10,11 Pesticides are responsible for oxidative stress that causes free radicals generation, leading to deoxyribonucleic acid (DNA) fragmentation.12,13
Lambda-cyhalothrin (LCT), a type II pyrethroid pesticide, is used worldwide to control pests in a variety of agricultural crops. LCT is chemically alpha-cyano-3phenoxybenzy3-(2-chloro-3,3,3 trifluoropropenyl)-2,2,dimethylcyclo- propane carboxylate. It revealed that lambda-cyhalothrin is moderately toxic for mammals12,13 and highly toxic for fish, aquatic invertebrates and bees. LCT at low concentrations can cause death in these species.14,15 The degree of concentration and nature of solvent are important for the toxicity of lambda cyhalothrin.16
Taurine (2-aminoethane sulphonic acid), a free intracellular sulfonated beta amino acid, is present in many animal tissues especially muscle, brain, liver, heart, etc.17 From the metabolism of methionine and cysteine, taurine is derived and is much more concentrated in pro-inflammatory cells such as polymorphonuclear phagocytes and in the retina.18 Taurine takes part in bile acid conjugation, detoxification, osmoregulation and modulation of cellular calcium level.19-21Taurine acts as major antioxidant in most living organism which attributes its ability to stabilize biomembranes17 and to scavenge reactive oxygen species.22
For any pesticide, the exposure concentration is important in the alteration of its toxicity. So in our present study, LCT were administered at different concentrations to find out the exact toxic dose levels of LCT which can produce hematological, hepatic and gonadal toxicity in male and female Wistar rat. At the same time, investigation was also carried out to evaluate the protective role of taurine against LCT induced alterations in lipid metabolism of female rat.
MATERIALS AND METHODS
Chemicals and Reagents
Lambda cyhalothrin 5% emulsifiable concentrate (EC) was procured from RPC Agro Industries, Kolkata. Taurine was purchased Sigma Aldrich Inc., USA. All other chemicals used were of analytical grade and were purchased from Merck India Ltd, Himedia India Ltd, etc.
Animals and Care
For the present study mature Wistar male and female albino rats (weighing 130-150 g) were taken and the animals were housed in polypropylene cages at an ambient temperature of 25 °C ±2 °C with 12 hrs light-dark cycle. The rats were acclimatized for one week prior to different treatments. The standard laboratory feed and water were supplied throughout the period of experimentation. The present study was approved by the Institutional Animal Ethical Committee (IAEC), registered under Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Govt. of India and performed in accordance to the relevant laws and guidelines of the CPCSEA.
Experimental Design
A commercial formulation of lambda cyhalothrin 5% EC ‘Karate’ was used for the study. Dilution of lambda cyhalothrin was done in distilled water to acquire the test concentrations. The test concentrations of lambda cyhalothrin were measured from the percentage of the active ingredient present in above mentioned commercial formulation of lambda cyhalothrin.
Healthy mature Wistar rats (n=6, for control and each dose group) of either sex selected by random sampling, were used for the study. The rats were kept fasting for overnight providing only sufficient water, after which LCT were administered orally for 14 consecutive days at the dose level of 6.89 mg/kg body wt. (i.e. 1/11th LD50 dose) for male and 5.15 mg/kg body wt. (i.e. 1/11th LD50 dose) for female. Sharma et al23 reported that oral LD50 dose of LCT for mature male and female rats were 75.85 and 56.69 mg/kg body wt. respectively. The procedure was repeated for the dose levels of 7.58(1/10th LD50), 8.42(1/9th LD50), 10.83(1/7th LD50), 15.17(1/5th LD50), 18.96(1/4th LD50), 25.28(1/3rd LD50) mg/kg body wt.24 for male rats. For female rats, lambda-cyhalothrin were administered orally at the dose levels of 5.66(1/10th LD50), 6.29(1/9th LD50), 8.09(1/7th LD50), 11.33(1/5th LD50), 14.17(1/4th LD50), 18.89(1/3rd LD50) mg/kg body weight. Dose solutions were freshly prepared immediately before usage.
In another separate set of experiment, female rats were divided into the following six groups each containing six to assess LCT induced toxicity to lipid metabolism and to find out the protective effect of taurine against this toxicity. Taurine was administered at the dose level of 50 mg/kg body wt., which was effectively used to protect the toxicity induced by various xenobiotics.25,26
1. Group A: Control (no treatment).
2. Group B: Taurine control (50 mg/kg body wt.).
3. Group C: LCT low dose (1/9 of LD50 value i.e., 6.3 mg/ kg body wt.).
4. Group D: Taurine (50 mg/kg body wt.)+lambda-cyhalothrin low dose (6.3 mg/kg body wt.).
5. Group E: Lambda-cyhalothrin high dose (1/5 of LD50 value i.e., 11.33 mg/kg body wt.).
6. Group F: Taurine (50 mg/kg body wt.)+lambda-cyhalothrin high dose (11.33 mg/kg body wt.).
At the end of the doses, the animals were fasted overnight. On 15th day, the rats were anesthetized with pentobarbital sodium (35 mg/kg) and sacrificed by cervical dislocation. Samples were collected and stored at -80 ºC until analysis.
Study on Body Weight
Body weight of individual overnight fasting male and female animals was taken regularly before the administration of LCT. After 24 hrs of the treatment of the last dose, all animals were weighed and then sacrificed.
Measurement of Haematological Parameters in Male Rat
Erythrocyte count:
Erythrocyte count was done by the dilution of blood with red blood cell (RBC) dilution fluid (1:200) and the total erythrocyte counts27 were expressed as ×106 /mm3 .
Estimation of haemoglobin percentage:
The haemoglobin percentage was measured by cyanmethemoglobin method.28 Using Drabkin’s diluent as a blank, the optical density was measured at 540 nm.
Total leukocyte count:
Blood was diluted (1:20) with white blood cell (WBC) dilution fluid and total leukocyte were counted using Neubaur haemocytometer.27
Study of Hepatic Biomarkers in Male Rat
Assay of serum glutamate-oxaloacetate transaminase (SGOT) and serum glutamate-pyruvate transaminase (SGPT):
For SGOT sample, 1 ml of buffer substrate (2.66 gm aspartic acid, 60 mg α-ketoglutaric acid and, 20.5 ml of 1(N) NaOH and 100ml of volume was made by 0.1 M phosphate buffer, pH-7.4) and for SGPT sample, 1 ml of buffer substrate (1.78 gm DL-alanine, 30 mg α-ketoglutaric acid, 20 ml of 0.1M phosphate buffer and 1.25 ml of 0.4(N) NaOH) were taken and waited for 5 min at 37ºC. Then 0.2 ml of serum sample was mixed and incubated at 37ºC for 60 min. To prepare standard, 0.2 ml of working standard (200 µM/100 ml) was taken in a test tube and 0.8 ml of buffer substrate was added. For blank, 1.0 ml of buffer substrate was taken. In each of sample, standard and blank test tubes, 1 ml of 2,4 dinitrophenylhydrazine hydrochloride (DNPH) solution were added and waited for another 20 min. Then 10 ml of 0.4(N) NaOH was mixed and waited for 10 minutes. Readings were taken at 520 nm in spectrophotometer29 (UV-245 Shimadzu, Japan).
Assay of hepatic malondialdehyde (MDA):
Malondialdehyde (MDA) was determined by the mixing of 1 ml of sample with 0.2 ml of 8.1% sodium dodecyl sulfate, 1.5 ml of acetate buffer (20%, pH-3.5) and 1.5 ml of aqueous solution of thiobarbituric acid (0.8%) and the mixtures were boiled for 60 min at 95 ºC. After heating when the red pigment was produced, that was extracted with 5 ml of n-butanol-pyridine (15:1) and centrifuged at 5000 rpm for 10 min at room temperature. The optical density of supernatants was measured at 535 nm.30
Estimation of hepatic reduced glutathione (GSH) content:
100 µl of sulfosalicylic acid was mixed with 200 μl of sample. Then the mixture was allowed for centrifugation at 3000 rpm for 10 min. With the supernatant, 1.8 ml of DTNB was included and shaken well.31 Final reading was noted at 412 nm.
Study of Male Reproductive Parameters
Measurement of seminal fructose concentration:
In a centrifuge tube, 1 ml of diluted seminal plasma (five times dilution was done by mixing 0.1 ml of seminal plasma with 4.9 ml of distilled water) was added with 0.3 ml of 1.8 gm% ZnSO4 and 2 ml of 0.1 M NaOH. After 15 min, the mixture was centrifuged at 2000 g to obtain the supernatant. Then seminal fructose concentration was measured by taking 0.5 ml of supernatant as sample, 0.5 ml of 0.14 mM and 0.28 mM fructose solutions as two standards and 0.5 ml of distilled water as blank. Then, 0.5 ml of indole reagent and 5 ml of concentrated HCl were added to each test tube. The test tubes were then incubated at 50 ºC for 20 min and were cooled in ice water and then in room temperature.32 The reading was taken at 470 nm in spectrophotometer (UV-245 Shimadzu, Japan).
Estimation of testicular cholesterol:
Testicular tissue was homogenized with 0.5% FeCl3 solution at a conc. of 20 mg/ml. Supernatant was collected after centrifugation of the homogenized tissue at 2000 rpm for 10 min. Then 0.1 ml of supernatant was added with 6 ml of glacial acetic acid to prepare sample. Simultaneously 5.9 ml of glacial acetic acid was added with 0.1 ml of working standard and 0.1 ml of distilled water to prepare standard. Blank was prepared by mixing 6 ml of glacial acetic acid and 0.1 ml of distilled water. Then 4 ml of colour reagents were added to each, mixed vigorously and stand for 20 minutes for spectrophotometric reading at 570 nm against blank.33
Assay of testicular malondialdehyde (MDA) and reduced glutathione (GSH) content:
Testicular malondialdehyde (MDA) and reduced glutathione (GSH) content were measured by the respective above mentioned methods.30,31
Study of Female Reproductive Parameters
Estimation of ovarian Δ5, 3β-hydroxysteroid dehydrogenase (Δ5, 3β-HSD) activity:
Homogenizing media was prepared by 20 ml of glycerol, 0.01 M EDTA in 0.05 M phosphate buffer in 100 ml with redistilled water. Tissue homogenate (20 mg/ml homogenizing media) was centrifuged at 10,000 rpm for 30 min at 4 ºC in a cold centrifuge. One ml of supernatant was mixed with 1 ml of sodium pyrophosphate buffer and 40 µl of dehydroepiandrosterone (DHEA). After addition of 0.1 ml of NAD, the activity of ∆5 ,3β-HSD was measured at 340 nm against a blank (without NAD).34
Assay of ovarian cholesterol, malondialdehyde (MDA) and reduced glutathione (GSH) content:
Ovarian cholesterol, malondialdehyde (MDA) and reduced glutathione (GSH) content were measured by the respective above mentioned methods.33,30,31
Study of the protective effect of taurine on serum bilirubin and lipid profile in female rat in selected dose levels
Serum bilirubin was determined by the method of Jendrassik and Grof using commercial diagnostic reagent kit.35 Serum cholesterol (CHO), triglyceride (TG) and high density lipoprotein cholesterol (HDL) were estimated by the commercial diagnostic reagent kit.36 VLDL was calculated using the formula (TG/5).37 LDL concentration (mg/dL) was estimated indirectly from the concentrations of CHO, TG and HDL using the equation37 LDL=CHO-(VLDL+HDL).
Statistical Analysis
The data was expressed as Mean±SEM. The differences between the means of each group were tested using a one way ANOVA test (using a statistical package, Origin 6.1, Northampton, MA, USA). p<0.05 was considered to indicate a statistically significant difference.
RESULTS
General Observations
After each treatment of lambda cyhalothrin, the animals were kept under observation at least once in 30 min interval. Special attention was given up to 4 hrs from the time of treatment. Rats those were exposed to different doses of lambda cyhalothrin over a period of 14 days produce ataxia, agitation, rolling and also tremors which were considered as the signs of toxicity up to eleventh day of treatment after which the symptoms were reduced.
Changes in Body Weight, Food and Water Intake
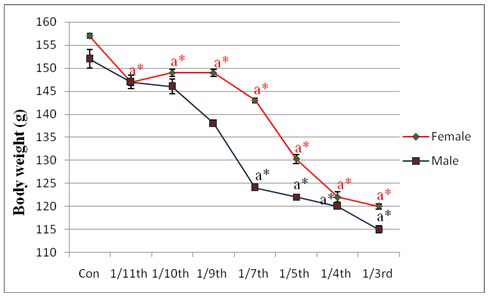
Significant changes in final body weight were found at the dose level of 10.83 mg/kg body wt. (1/7th LD50) in male rats and at the dose level of 5.15 mg/kg body wt. (1/11th LD50) in female rat (Figure 1). No differences in food and water consumption were seen in last consecutive four days of treatment in the experimental schedule but there was little non-significant alterations observed in food consumption at the exposure dose levels of 1/4th and 1/3rd LD50.
Figure 1: Effect of lambda cyhalothrin on body weight of male and female rat. Results are expressed as Mean±SEM. Analysis is done by one way ANOVA. Superscript a, control group versus all other groups (*indicates p<0.001). (For male and female the LD50 values are different which are mentioned in materials and methods section).
Effect on Haematological Parameters in Male Rat
To find out the dose dependent effect of LCT, some clinically significant biochemical parameters from each system were studied. From the study it was seen that total erythrocyte count was decreased significantly (p<0.05) from the dose level of 10.83 mg/kg body wt. (1/7th LD50)to 25.28 mg/kg body wt. (1/3rd LD50) in male rat (Table 1). No significant changes were found below 10.83 mg/kg body wt. (1/7th LD50 dose). Maximum toxic effect of LCT was exhibited at the 15.17 mg/kg body wt. (1/5th LD50 dose) without any mortality. Interestingly, the rate of mortality was increased from the dose level of 18.96 mg/kg body wt. (1/4th LD50). Similar results were found in case of haemoglobin
percentage (p<0.01). Table 1 displays the significant (p<0.001) increase in leukocyte count in lambda cyhalothrin treated male rats from the dose level of 1/7th LD50 to 1/3rd LD50.
Table 1: The effect of lambda cyhalothrin on haematological parameters in male rat. Results are expressed as Mean±SEM. Analysis is done by one way ANOVA. Superscript a, Control group versus all other groups (*indicates p<0.05,**indicates p<0.01, ***indicates p<0.001).
|
Group
|
Erythrocyte count (×106 / mm3 ) |
Hb percentage (gm/dL) |
Leukocyte count (×106 /µl)
|
| Control |
7±0.3
|
14±0.5 |
6±0.17
|
| 1/11th LD50 dose (6.89 mg /kg body wt.) |
7.2±0.2
|
14±1.1 |
6±0.11
|
| 1/10th LD50 dose (7.58 mg /kg body wt.) |
7±0.1
|
13.3±1.4 |
6.03±0.14
|
| 1/9th LD50 dose (8.42 mg /kg body wt.) |
7±0.06
|
13.6±0.6 |
6.03±0.15
|
| 1/7th LD50 dose (10.83 mg /kg body wt.) |
6±0.2a*
|
10±0.6a** |
8±0.11a***
|
| 1/5th LD50 dose (15.17 mg /kg body wt.) |
5.9±0.1a*
|
8±0.6a** |
9.2±0.12a***
|
| 1/4th LD50 dose (18.96 mg /kg body wt.) |
5.9±0.2a*
|
8±0.6a** |
9.1±0.17a***
|
| 1/3rd LD50 dose (25.28 mg /kg body wt.) |
5.9±0.1a*
|
8±1.1a** |
9.2±0.12a***
|
Effect on Hepatic Biomarkers in Male Rat
Activities of serum glutamate-oxaloacetate transaminase (SGOT) and serum glutamate-pyruvate transaminase (SGPT):
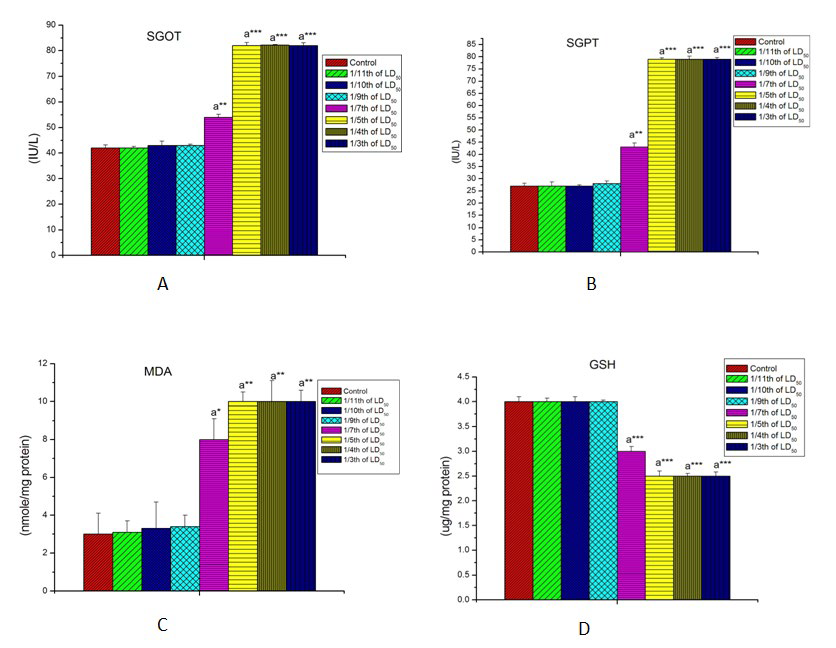
The effects of LCT on SGOT, SGPT are shown in Figures 2A, and 2B respectively. The activity of these two important hepatic transaminase enzymes were increased significantly (p<0.001) with increase in the concentration of LCT as compared to the control rats. No significant alterations were noted below 10.83mg/kg body wt. (1/7th LD50 dose) in male rat.
Figure 2: Effect of lambda cyhalothrin on some hepatic biomarkers in male rat. A= Effect on SGOT. B= Effect on SGPT. C=Effect on liver MDA. D=Effect on liver GSH. Results are expressed as Mean±SEM. Analysis is done by one way ANOVA. Superscript a, Control group versus all other groups (*indicates p<0.05, **indicates p<0.01, ***indicates p<0.001).
Hepatic lipid peroxidation and glutathione content:
Hepatic malon-di-aldehyde (MDA) and reduced glutathione content in the control and experimental groups of male rats are shown in Figures 2C and 2D. MDA was found to be significantly increased (p<0.01) whereas reduced glutathione content was decreased significantly (p<0.001) in a dose-dependent manner from the dose level of 10.83 mg/kg body wt. (1/7th LD50 dose) onwards.
Effect on Male Reproductive System
Effect of LCT on seminal fructose concentration and testicular cholesterol:
As shown in Table 2, the LCT induced reproductive toxicity by reducing seminal fructose concentration (p<0.001) and elevating testicular cholesterol level (p<0.05).
Table 2: Shows the effect of lambda cyhalothrin on some male reproductive parameters. Results are expressed as Mean±SEM. Analysis is done by one way ANOVA. Superscript a, control group versus all other groups (*indicates p<0.05, **indicates p<0.01, ***indicates p<0.001)
|
Group
|
Seminal fructose concentration (nmole of fructose/lit of seminal plasma) |
Testicular Cholesterol (mg/gm) |
Testicular MDA (nmole/mg protein) |
Testicular GSH (µg/mg protein)
|
| Control |
20±0.3 |
1.4±0.1 |
2.5±0.2 |
3.5±0.03
|
| 1/11thLD50 |
20±0.6
|
1.4±0.2 |
2.6±0.2 |
3.4±0.12
|
| 1/10th LD50 |
20±0.5 |
1.4±0.05 |
2.6±0.1 |
3.4±0.03
|
| 1/9th LD50 |
19.5±0.2
|
1.4±0.1 |
2.6±0.3 |
3.4±0.09
|
| 1/7th LD50 |
18±0.1a**
|
1.9±0.1a* |
4±0.2a* |
2.9±0.12a**
|
| 1/5th LD50 |
15±0.3a***
|
2.4±0.3a* |
5±0.3a** |
2.36±0.22a***
|
| 1/4th LD50 |
15±0.2a***
|
2.4±0.1a* |
5±0.2a** |
2.36±0.25a***
|
| 1/3rd D50 |
15±0.4a***
|
2.4±0.05a* |
5±0.5a** |
2.36±0.24a***
|
Impact of LCT on testicular oxidative stress and antioxidant status:
Table 2, also shows the effect of LCT on MDA and GSH. Testicular MDA was found to be increased significantly (p<0.01) in response to LCT treatment whereas a significant decrease (p<0.001) in testicular GSH level was also noted. Significant changes were detected from the dose level of 10.83 mg/kg body wt. (1/7th LD50 dose) onwards.
Effect on Female Reproductive Function
Ovarian steroidogenic key enzyme activities and cholesterol content:
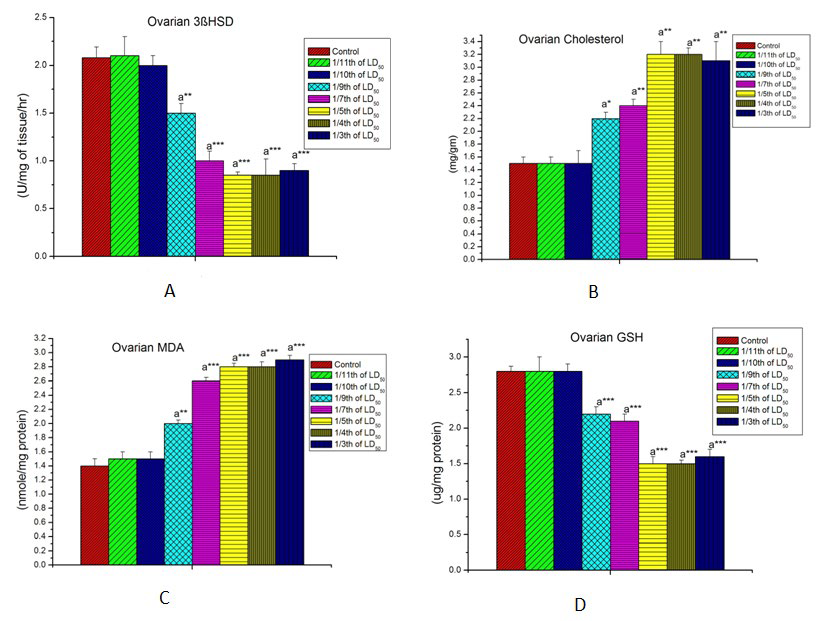
LCT induced ovarian toxicity was exhibited by significant diminution in ovarian steroidogenic enzyme activities and elevation in ovarian cholesterol content at the dose level of 6.29 mg/kg body wt. (1/9th LD50 dose) and were continued towards increased dose levels of LCT (Figures 3A and 3B).
Figure 3: Effect of lambda cyhalothrin on female reproductive biomarkers. A=Effect on ovarian 3β HSD. B= Effect on ovarian cholesterol. C= Effect on ovarian MDA. D=Effect on ovarian GSH. Results are expressed as Mean±SEM. Analysis is done by one way ANOVA. Superscript a, control group versus all other groups (*indicates p<0.05, **indicates p><0.01, ***indicates p><0.001).
Effect of LCT in malondialdehyde (MDA) and reduced glutathione (GSH) level:
LCT intoxicated rats shows a marked dose-dependent increase (p<0.001) in the lipid peroxidation, specified in term of MDA (Figure 1). Decline in GSH levels in LCT treated rat may also an indication of oxidative stress as GSH is used for the detoxification of reactive toxic substances resulted from LCT exposure at the dose level of 6.29 mg/kg body wt. (1/9th LD50 dose) (Figures 3C and 3D).
Selection of Experimental Dose of Lambda Cyhalothrin
From these above findings for male rats, 10.83(1/7th LD50 dose) and 15.17(1/5th LD50 dose) mg/kg body wt. and for female rats, 6.29 mg/kg body wt. (1/9th LD50 dose) and 11.33(1/5th LD50 dose) mg/kg body wt. were selected as effective doses for our further studies. At the 1/7th LD50 dose, significant alterations were seen in different systemic biochemical parameters of male rat. In female rat first significant toxic response was found at the dose level of 6.29 mg/kg body wt. (1/9th LD50 dose). So in case of male and female rat, 1/7th LD50 dose and 1/9th LD50 dose were considered as effective low dose. Better toxic response was found at 1/5th LD50 dose for both male and female rat after which animal mortality was increased. So this dose was considered as effective high dose for future studies.
Effect on Lipid Profiles in Female Rat
The data from Table 3 reflected marked alterations in the levels of serum total cholesterol, lipoproteins and bilirubin. The total cholesterol concentration was found to be significantly increased (p<0.001) in the LCT treated group compared to control. Serum triglyceride, VLDL-cholesterol and LDL-cholesterol in the LCT treated rats were also found to be elevated when compared to normal rats where as HDL was markedly (p<0.001) reduced in LCT treated female rats. Our results also showed a significant increase of serum bilirubin level in LCT intoxicated rats. Pre treatment with taurine has caused the significant alleviation in serum total cholesterol, LDL cholesterol and HDL cholesterol in LCT treated rats. Pretreatment with taurine also improved the altered serum triglyceride and bilirubin level (Table 3).
|
Parameters
|
Group-A |
Group-B |
Group-C |
Group-D |
Group-E |
Group-F
|
|
Serum total Cholesterol (mg/dL)
|
120±0.7
|
112±1.5a*** |
138±0.8a*** |
127±1.3a***b*** |
149±1.5a*** |
137±1.2a***c***
|
|
Serum triglyceride (TG) (mg/dL)
|
98±1.0
|
92±1.0a** |
104±1.5a*c*** |
98±0.5b* |
121±1.0a*** |
104±1.5a*c***
|
|
High density lipoprotein (HDL) (mg/dL)
|
45±0.7
|
51±1.0a*** |
38±0.2 a*** |
47±1.2b*** |
29±1.0a*** |
37±0.5 a***c*** |
|
Very low density lipoprotein (VLDL) (mg/dL)
|
19.6±0.2
|
18.4±0.2 |
21.6±0.1a*** |
20±0.3b* |
24.2±0.2a*** |
20.8±0.4a**c***
|
|
Low density lipoproteins (LDL) (mg/dL)
|
55.3±1.8
|
42.6±2.7a** |
78.4±0.8a*** |
60±0.2a*b*** |
95.8±0.7a*** |
79.2±1.4a**c***
|
|
Serum bilirubin (mg/dL)
|
0.3±0.05
|
0.4±0.06 |
1±0.07a*** |
0.5±0.02a**b*** |
1.7±0.1a*** |
1.2±0.1a***c**
|
DISCUSSION
The present study was carried out to assess the toxic effects of LCT on the different system of male, female Wistar rats, and also to search out the alleviating role of taurine under this toxic condition. In toxicological studies, body weight is a basic benchmark for evaluation of organ toxicity. In the present study, oral administration of LCT brought about a significant reduction in body weight of both male female rats.
A significant change in erythrocyte counts, haemoglobin percentage and leukocyte were detected in lambda cyhalothrin exposed rats and these pointed out the physiological disruption in the rat haemopoietic system. Haemolysis of blood cells38 may cause the decrease in erythrocyte counts in LCT intoxicated rats which in turn tends to be responsible for the reduction in haemoglobin percentage. In the present study, decreased biosynthesis of haem in bone marrow may also lead to the significant reduction in haemoglobin percentage. Increased leukocyte in lambda cyhalothrin treated group may arise due to the immediate activation of the immune system of the body39 against lambda cyhalothrin.
SGOT, SGPT are two important hepatic enzyme biomarkers of hepatotoxicity. In the present study a significant increase in SGOT, SGPT level after LCT treatment at different concentration point out towards active utilization of amino acids in energy-yielding metabolic processes like gluconeogenesis. Pyrethroids induced oxidative stress by the elevation of lipid peroxidation products.40,41 Elevated MDA level in LCT intoxicated rat liver was in an agreement with the above statement. This type of result also suggested that LCT produces hepatic injury and pathogenesis through the generation of free radicals and by the alteration of antioxidant system. Decrease in cellular GSH concentrations may be through low production or non-enzymatic oxidation of GSH to glutathione disulfide (GSSG) due to oxidative stress in LCT treated rat liver at different dose levels.
Here the results also reflect the male reproductive dysfunction after LCT exposure at different dose levels. The reduction in fructose content in seminal fluid collected from LCT intoxicated rats were drawn attention towards the secretory ability of seminal vesicles and the nutritive potential for the semen. Previous studies42,43,44 reported that ROS were involved in the toxicity of various pesticides. ROS inhibits steroidogenesis by disrupting cholesterol transport to mitochondria.45 LCT elevated testicular cholesterol at the dose level of 10.83 mg/kg body wt. (1/7th LD50 dose) and also on above dose levels. An increase in MDA, the mostly used biomarker of lipid peroxidation, indicates serious cellular damage, inhibition of several enzymes and cellular functions.46 GSH, one of the most potent biological molecules, play a key role in the detoxification of the reactive toxic metabolites. A considerable decline in GSH levels in liver LCT treated rat may be due to its utilization to challenge the common oxidative stress.
According to the results, increase in ovarian cholesterol and significant decrease in ovarian steroidogenic enzyme activity pointed out towards the LCT induced ovarian toxicity through diminishing steroidogenesis. The maintenance of high redox potential is a prerequisite for assuring the reproductive system functions in a healthy organism.47,48 Increased ovarian MDA and decreased ovarian GSH level in LCT intoxicated rats compared to control may be an indication of oxidative stress due to LCT exposure at the dose level of 6.29 mg/kg body wt. (1/9th LD50 dose) and also on above dose levels.
The elevation in serum total cholesterol level in female rats was observed in the LCT treated groups indicating the hypercholesterolemic action of LCT due to blockage of liver bile ducts causing reduction or cessation of its secretion to the duodenum.49 Pretreatment of taurine by its hypocholesterolemic action caused reduction in serum total cholesterol. Elevated serum triglyceride, VLDL-cholesterol and LDL-cholesterol with reduction of HDL in LCT treated rats may suggest that LCT has strong toxic potential to alter normal body physiology. These may be attributed to the increased fat catabolism in response to LCT.50 Our results also showed a significant increase of serum bilirubin level in LCT intoxicated rats. The increased level of total bilirubin in treated female rats is an indicator of hyper-bilirubinemia, a useful indicator of hepatocellular dysfunction.51,52 Pretreatment with taurine improved the altered lipid profile and bilirubin level in female rats. Taurine play a role in conjugation reaction with bile acids in the liver. Consequently, bile acid synthesis is increased53 by simultaneous rise in the messenger RNA (mRNA) expression and activity of cholesterol 7α-hydroxylase, a rate-limiting enzyme in hepatic bile acid synthesis.54 The primary mechanisms responsible for the hypocholesterolemic action of taurine may be due to the increased conversion of cholesterol into bile acids through the stimulation of cholesterol 7α-hydroxylase enzyme activity.
CONCLUSION
The present study showed that LCT exposure produced hematological, hepatic and gonadal toxicity at 10.83 mg/kg body wt. (1/7th LD50 dose) in male and at 6.29 mg/kg body wt. (1/9th LD50 dose) in female rat and also on above dose levels. Taurine has the potential to mitigate LCT induced altered lipid metabolic biomarkers in female rat.
ACKNOWLEDGMENT
The authors are thankful to the authority of Vidyasagar University, Midnapore, India for providing all the facilities to execute this study.
CONFLICTS OF INTEREST
The authors declare that they have no conflicts of interest.








