1. Karl TR, Trenberth KE. Modern global climate change. Science. 2003; 302(5651): 1719-1723. doi: 10.1126/science.1090228
2. Roth MS, Deheyn DD. Effects of cold stress and heat stress on coral fluorescence in reef-building corals. Sci Rep. 2013; 3: 1421. doi: 10.1038/srep01421
3. Melillo JM, McGuire AD, Kicklighter DW, Moore B, Vorosmarty CJ, Schloss AL. Global climate change and terrestrial net primary production. Nature. 1993; 363(6426): 234-240. doi: 10.1038/363234a0
4. Abu-Dieyeh Z. Effect of chronic heat stress and long-term feed restriction on broiler performance. Int J Poult Sci. 2006; 5(2): 185-190. doi: 10.3923/ijps.2006.185.190
5. Prieto M, Campo J. Effect of heat and several additives related to stress levels on fluctuating asymmetry, heterophil: Lymphocyte ratio, and tonic immobility duration in White Leghorn chicks. Poult Sci. 2010; 89(10): 2071-2077. doi: 10.3382/ps.2010-00716
6. St-Pierre N, Cobanov B, Schnitkey G. Economic losses from heat stress by US livestock industries. J Dairy Sci. 2003; 86: E52- E77. doi: 10.3168/jds.S0022-0302(03)74040-5
7. Borges SA, Fischer da Silva AV, Ariki J, Hooge DM, Cummings KR. Dietary electrolyte balance for broiler chickens exposed to thermoneutral or heat-stress environments. Poult Sci. 2003; 82(3): 428-435. doi: 10.1093/ps/82.3.428
8. Mohanaselvan A, Bhaskar E. Mortality from non-exertional heat stroke still high in India. Int J Occup Environ Med. 2014; 5(4): 224.
9. Lara LJ, Rostagno MH. Impact of heat stress on poultry production. Animals. 2013; 3(2): 356-369. doi: 10.3390/ani3020356
10. Salzman NH. Microbiota-immune system interaction: An uneasy alliance. Curr Opin Microbiol. 2011; 14(1): 99-105. doi: 10.1016/j.mib.2010.09.018
11. Elson CO, Cong Y. Host-microbiota interactions in inflammatory bowel disease. Gut Microbes. 2012; 3(4): 332-344. doi: 10.4161/gmic.20228
12. Salminen S, Isolauri E. Intestinal colonization, microbiota, and probiotics. J Pediatr. 2006; 149(5): S115-S120. doi: 10.1016/j. jpeds.2006.06.062
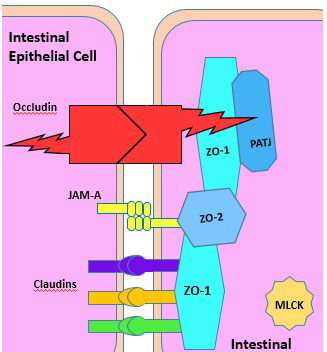
13. Cummins PM. Occludin: One protein, many forms. Mol Cell Biol. 2012; 32(2): 242-250. doi: 10.1128/MCB.06029-11
14. Di Pierro M, Lu R, Uzzau S, et al. Zonula occludens toxin structure-function analysis Identification of the fragment biologically active on tight junctions and of the zonulin receptor binding domain. J Biol Chem. 2001; 276(22): 19160-19165. doi: 10.1074/jbc. M009674200
15. Assimakopoulos SF, Papageorgiou I, Charonis A. Enterocytes’ tight junctions: From molecules to diseases. World J Gastrointest Pathophysiol. 2011; 2(6): 123. doi: 10.4291/wjgp.v2.i6.123
16. Hu YJ, Wang YD, Tan FQ, Yang WX. Regulation of paracellular permeability: Factors and mechanisms. Mol Biol Rep. 2013; 40(11): 6123-6142. doi: 10.1007/s11033-013-2724-y
17. Awad W, Bohm J, Razzazi-Fazeli E, Ghareeb K, Zentek J. Effect of addition of a probiotic microorganism to broiler diets contaminated with deoxynivalenol on performance and histological alterations of intestinal villi of broiler chickens. Poult Sci. 2006; 85(6): 974-979.
18. He H, Kogut MH. CpG-ODN-induced nitric oxide production is mediated through clathrin-dependent endocytosis, endosomal maturation, and activation of PKC, MEK1/2 and p38 MAPK, and NFkappaB pathways in avian macrophage cells (HD11). Cellular signalling. 2003; 15(10): 911-917. doi: 10.1016/S0898-6568(03)00100-1
19. Murakami T, Felinski EA, Antonetti DA. Occludin phosphorylation and ubiquitination regulate tight junction trafficking and vascular endothelial growth factor-induced permeability. J Biol Chem. 2009; 284(31): 21036-21046. doi: 10.1074/jbc.M109.016766
20. Qin L, Huang W, Mo X, Chen Y, Wu X. LPS induces occludin dysregulation in cerebral microvascular endothelial cells via MAPK signaling and augmenting MMP-2 levels. Oxid Med Cell Longev. 2015; 2015. doi: 10.1155/2015/120641
21. Muthusamy A, Lin CM, Shanmugam S, Lindner HM, Abcouwer SF, Antonetti DA. Ischemia-reperfusion injury induces occludin phosphorylation/ubiquitination and retinal vascular permeability in a VEGFR-2-dependent manner. J Cereb Blood Flow Metab. 2014; 34(3): 522-531. doi: 10.1038/jcbfm.2013.230
22. Suzuki T, Elias BC, Seth A, et al. PKC eta regulates occludin phosphorylation and epithelial tight junction integrity. Proc Natl Acad Sci USA. 2009; 106(1): 61-66. doi: 10.1073/pnas.0802741106
23. Hirase T, Kawashima S, Wong EY, et al. Regulation of tight junction permeability and occludin phosphorylation by Rhoap160ROCK-dependent and -independent mechanisms. J Biol Chem. 2001; 276(13): 10423-10431. doi: 10.1074/jbc.M007136200
24. Murakami T, Frey T, Lin C, Antonetti DA. Protein kinase C$\beta$ phosphorylates occludin regulating tight junction trafficking in vascular endothelial growth factor-induced permeability in vivo. Diabetes. 2012; 61(6): 1573-1583. doi: 10.2337/db11-1367
25. Al-Sadi R, Ye D, Dokladny K, Ma TY. Mechanism of IL-1$\beta$-induced increase in intestinal epithelial tight junction permeability. J Immunol. 2008; 180(8): 5653-5661. doi: 10.4049/jimmunol.180.8.5653
26. Turner JR, Angle JM, Black ED, Joyal JL, Sacks DB, Madara JL. PKC-dependent regulation of transepithelial resistance: Roles of MLC and MLC kinase. Am J Physiol. 1999; 277(3 Pt 1): C554-C562.
27. Turner JR, Rill BK, Carlson SL, et al. Physiological regulation of epithelial tight junctions is associated with myosin light-chain phosphorylation. Am J Physiol. 1997; 273(4 Pt 1): C1378-C1385.
28. Bosenberg AT, Brock-Utne JG, Gaffin SL, Wells MT, Blake GT. Strenuous exercise causes systemic endotoxemia. J Appl Physiol. 1988; 65(1): 106-108.
29. Ilan Y. Leaky gut and the liver: A role for bacterial translocation in nonalcoholic steatohepatitis. World J Gastroenterol. 2012; 18(21): 2609. doi: 10.3748/wjg.v18.i21.2609
30. Baxter MF, Merino-Guzman R, Latorre JD, et al. Optimizing fluorescein isothiocyanate dextran measurement as a biomarker in a 24-h feed restriction model to induce gut permeability in broiler chickens. Front Vet Sci. 2017; 4. doi: 10.3389/fvets.2017.00056
31. Yan Y, Kolachala V, Dalmasso G, et al. Temporal and spatial analysis of clinical and molecular parameters in dextran sodium sulfate induced colitis. PLoS One. 2009; 4(6): e6073. doi: 10.1371/journal.pone.0006073
32. Varasteh S, Braber S, Akbari P, Garssen J, Fink-Gremmels J. Differences in susceptibility to heat stress along the chicken intestine and the protective effects of galacto-oligosaccharides. PLoS One. 2015; 10(9): e0138975. doi: 10.1371/journal.pone.0138975