INTRODUCTION
According to World Health Organization (WHO), physical activity (PA) is related to movements of the body produced by skeletal muscles with an expenditure of energy.1 PA includes exercise as well as other activities, which involve bodily movement and are done as part of playing, working, active transportation, house chores and recreational activities.1
It is important to verify the difference between “physical activity” and “exercise”. Exercise is a type of PA that is planned, structured, repetitive and purposeful aiming at the improvement or maintenance of one or more components of physical fitness.2
Warburton and Bredin2 showed in a systematic review that the health benefits of exercise are evident, physical inactivity has been identified as the fourth leading risk factor for mortality in the world (6% of deaths worldwide).1 Some diseases have been correlated with physical inactivity such as metabolic syndrome (MetS), type 2 diabetes mellitus (DM2), stroke and obesity3 and, according to Rui et al4 a highly prevalent and expensive disease. Increasing PA is a social problem and not just an individual problem. It therefore requires a population-based, multi-sectoral, multidisciplinary and culturally relevant approach.1
Regular PA leads to enhanced energy consumption and is associated with reduced risk of prevalent diseases such as obesity, T2D Mand MetS.3 Studies indicate that combined diet and PA promotion programs can also prevent T2D Mand arterial hypertension and other conditions that can increase the risk to cardiovascular diseases.5 Arterial hypertension is a chronic disease that affects approximately 40% of the population, with higher incidence at older ages.6 Arterial hypertension is a risk factor for other cardiovascular diseases, such as heart failure, stroke, atherosclerosis as well as chronic renal disease.7 It is estimated that more than 50% of deaths from coronary diseases and stroke occur in hypertensive patients.8 In this context, the development of nonpharmacological therapies is a cost-effective strategy with few side effects, that helps in the prevention of comorbidities, that increase the cardiovascular risk of the patient. Carr et al9 reported that in a large populationbased case-control study was identified as a combination of lifestyle factors that appears to reduce risk of colorectal cancer, regardless of the patient’s genetic profile. These results reinforce the importance of primary prevention of colorectal cancer. Among non-pharmacological strategies to prevent cardiovascular disease and cancer, the physical exercise deserves consideration.
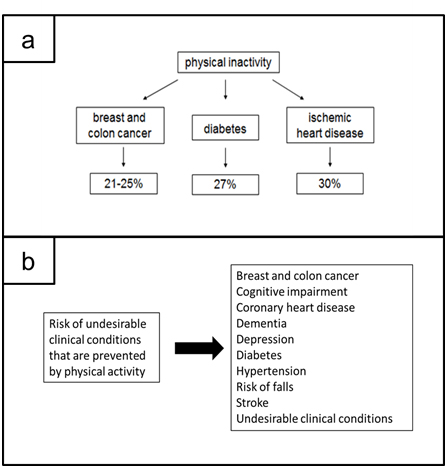
In Figure 1a are estimated at approximately the main undesirable causes due to the physical inactivity to the human beings, as suggested by Warburton and Bredin.2
Regular and adequate levels of PA in adults (i) reduce the risk of various undesirable clinical conditions, as it is shown in Figure Ib, (ii) improve bone and functional health; and (iii) are a key determinant of energy expenditure, and thus, it would be fundamental to energy balance and weight control.10–12
Figure 1. a. Main Undesirable Causes Due to the Physical Inactivity to the Human Beings; b. Risk of Undesirable Clinical Conditions that are Prevented by Physical Activity
There are various types of physical exercises, that, in general, can be divided into aerobic (AeE) and anaerobic (AnE).13 Following the American College of Sports Medicine (ACSM),14 AeE exercise involves large muscle groups, that can be maintained continuously and is rhythmic in nature. Cycling, dancing, walking, running, swimming and walking have considered as AeE. Studies have shown that AeE improves lipid profile, particularly by increasing HDL-C, in addition to having a positive impact on cardiovascular health, and the cortisol regulation.14 ACSM considers AnE an intense PA with short time duration. AnE has as energetic substrate, no more the oxygen, but the adenosine triphosphate via glycolysis and fermentation.14 Although with particular differences, both kind of exercises, (AeE and AnE) may exert beneficial influence on the cardiovascular system, besides to bring benefits to the lipid metabolism, the increase of the lean mass, and the increase of the basal metabolism.12
Among the wide variety of exercises, there is the whole body vibration exercise (WBVE), which is one of the types of AeE,15 induces hormonal and immunological responses. WBVE has also been shown to be efficient for hormonal responses. Evidence suggests that the combination of WBVE and resistance work may amplify the hormonal and immune responses due to increased neuromuscular load16 and as a response of the PA, the concentration of several hormones is altered, as the cortisol.
CORTISOL
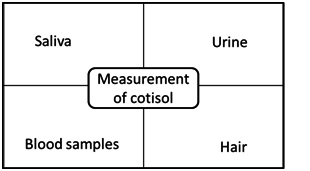
Cortisol is a glucocorticoid hormone (a steroid hormone), produced by the upper adrenal gland secreted by the adrenal gland cortex in response to conditions stressors. The hypothalamicpituitary-adrenal (HPA) axis is required for adaptation to stress. Moreover, the activation of the HPA axis leads to secretion of glucocorticoids, which act on multiple organ systems to redirect energy resources to meet actual or anticipated demand once secreted, cortisol is absorbed by a variety of tissues throughout the body, such as skeletal muscle. The presence cortisol mediates physiological processes that help in exercise capacity and tissue regeneration. It is described that cortisol possesses a diurnal pattern and thus collection timing is critical. The cortisol potentially affects the metabolism of lipids and carbohydrates. The high-level of this hormone is related with the development of undesirable clinical conditions (central adiposity, insulin resistance and hyperglycemia, besides hypertension and dyslipidemia).15,17,18 Wright et al15 have reported different specimens used to determination of a concentration of the cortisol in the body, as it is shown in Figure 2.
Figure 2. Reported Different Specimens Used to Determination of Concentration of the Cortisol
HAIR
Hair plays no vital role for humans, but its loss may be associated with several conditions (physiological, psychological, idiopathic, aging, or genetic predisposing complications).15 As hair can have a relevant impact on the quality of life and self-esteem of individuals, its loss can generate distress and suffering of some diseases and infections. Nevertheless, the hair loss can be consequence of metabolic and hormonal dysfunctions or stress.19 Figure 3 shows the main functions of the hair by Buffoliet al.20 It has also a sensory function, increasing the perception of the skin surface for tactile stimuli, and sub-serves important roles in sexual and social communication. In addition, authors have reported that hair can be used as a specimen to quantify the concentration of cortisol in the body (Figure 1b).10,11
Figure 3. Main Functions of the Hair by Buffoli et al20
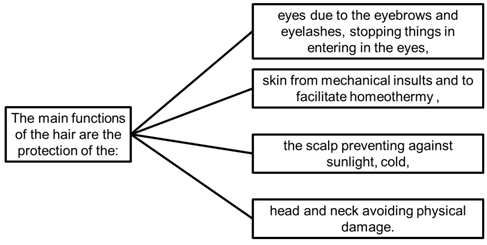
According to Pragst and Balikova,21 hair grows on average one centimeter per month. Among some important hair roles are skin protection against aggressors, thermoregulation, some specialized functions such as eyebrows and eyelashes. As the hair germ extends, the epithelium of the hair bulb encapsulates the dermal condensate to form the dermal papillae. Eventually, the layers form in the outer root sheath, the inner root sheath, the cuticle of the hair shaft, the cortex, and the medulla. Besides the hair follicle, sebaceous gland and the apocrine gland are also found.
It is relevant to point out the use of the hair at forensic studies and doping research. Considering, then, the need to develop a form of chronic stress measurement, the capillary measurement of cortisol was discovered and validated by Raul et al.22 In addition, there is chronic exposure to the stressor, with an important effect of exposure to stressors in capillary cortisol concentrations. This reveals deregulation of the HPA axis.23 The exposure to even a short stress can stimulate the HPA axis and can increase the level of cortisol that can persist up to 1 hour after exposure. Although adaptable in the short-term, repeated and chronic HPA axis stimulation and cortisol release may lead to allostatic loading.23
Putting together all the previous considerations, the aim of this narrative review is to present scientific information about the use of the hair to evaluate the concentration of cortisol in individuals that have performed PA.
HAIR CORTISOL AS A RETROSPECTIVE LEVEL OF PHYSICAL ACTIVITY
Hair cortisol is a proxy measure to the total retrospective activity of the HPA axis over the preceding months, much like hemoglobin A1c is a proxy measure of glucose control over the past 3-months.24 It is pointed out that hair cortisol analysis advances the science of aging by permitting the characterization of chronic stress as a risk factor for chronic illness progression and as a biomarker of the effectiveness of stress reduction interventions.15
Cortisol obtained from scalp hair is a lipophilic substance that originates from the vascular supply which nourishes the hair shaft follicular cells. The source of cortisol within the hair shaft is from circulation to the medullary region in the core of the hair.25 Sources of cortisol on the surface of the hair include both sweat as well as sebaceous glands which are most likely eliminated by the washing steps prior to grinding the hair for extraction. It is considered that the cortisol measured using the standard wash, grind, and extraction method most likely reflects free cortisol and not the bound steroid.7
Although controversial studies report that collection would be performed, it is understood that (i) the medullary region of the passive diffusion of blood, in which is the free cortisol fraction and (ii) cortisol the external cuticle of the secretions present in sebum or sweat.22
According to Ito et al26 hair follicles contain a functional equivalent of the HPA axis and can synthesize cortisol after stimulation of corticotrophin releasing hormone (CRH). For this reason, it is possible that data collection can be influenced by the cortisol concentrations produced in the hair by secretion in sebum or sweat, and not by the concentration of free cortisol.27
Several publications were accessed in a search in the database PubMed September 12th, 2018 using the keywords “hair” and “physical activity” and “cortisol”. They are indicated in Table 1, in which are presented the aim of the study and the findings of each study.
| Table 1. Publications Accessed in the Database PubMed with the Keywords “Hair” and “Physical Activity” and “Cortisol” |
| Studies |
Aim |
Findings |
| Teychenne et al28 |
To verify the association between common types of sedentary behavior (TV viewing, computer use, or overall sitting time) and objectively-measured stress, measured by hair cortisol concentration (HCC) amongst women living in socioeconomically disadvantaged neighbourhoods. |
It was shown that there was no association between any type of sedentary behaviour and HCC in either crude or adjusted models. Sedentary behaviour may not be linked to HCC (stress) in women living in socioeconomically disadvantaged neighbourhoods. |
| Hu et al29 |
To examine possible relations between HCC as a biomarker of chronic stress and childhood overweight and obesity. |
HCC had a significant relation with overweight and obesity in 6-9 years old childhood especially among girls. |
| Ullmann et al30 |
To try to identify relationships between PA, mental burden(s), subjective stress perceptions, depressiveness, anxiety, physical complaints, sense of coherence, resilience, and the long-term integrated steroid hormone levels in hair. |
The HCC, cortisone, and dehydroepiandrosterone were significantly correlated to mental and PA stress as well as to subjective stress perception. It was concluded that steroid concentrations in hair are decisive predictors for an increase in the long-term-HPA axis activity. Moreover, this biomarker is suitable for capturing the stress level after burdening events and PA. |
| Gerber et al31 |
To verify if HCC was associated with perceived stress (PS) and depressive symptoms in a sample of young adults and tested if HCC could explain the variation in depressive symptoms beyond PS before and after controlling vigorous PA levels |
Students with high HCC tend to report lower depressive symptoms and less PS. Increased perceived stress was associated with major depressive symptoms, and both cortisol and PS predicted depressive symptoms after vigorous PA control. Current data suggest that high HCC do not necessarily pose a health risk. The measurement of HCC can serve as a non-invasive and painless biomarker of chronic stress and mental disorders. |
| Gerbe et al32 |
To verify relationship between moderate PA and vigorous PA levels and HCC after taking into account age, gender, and PS. |
Moderate PA was not correlated with HCC. A significant correlation was found between vigorous PA and HCC. A regression analysis revealed that participants with higher vigorous PA had elevated HCC even after taking into account age, gender and PS. |
| HCC-hair cortisol concentration, PA-physical activity, HPA–hypothalamus-pituitary-adrenal, PS-perceived stress. |
DISCUSSION
The study of capillary cortisol presents a unique opportunity to evaluate chronic changes in cortisol concentrations in epidemiological studies. Wosu et al33 have investigated the assessment of cortisol concentrations in hair is one of the latest innovations to measure long-term exposure to cortisol.
Studies have indicated that in hair is possible to find specific steroid hormones. Moreover, researches have analyzed exogenous steroids and androgens for doping.31,32 Furthermore, works relating hormones, especially glucocorticoids are conducted in tests to assess the level of stress.34 Despite the increasing use of capillary analyzes, the detection of steroid hormones in hair has received relatively minor attention. The measurement of cortisol in human hair can be also done by high-performance liquid chromatography with fluorescence detection (HPLC-FLU) along with gas chromatographic mass spectrometry (GC-MS).35
Hair-corrected cortisol may be a chronic stress biomarker, as it evaluates retrospective stress, and does not require repeated measures for this, reducing costs on biochemical reagents. It would be also potentially useful in clinical and sporting contexts. Physiological responses to stress are often difficult to measure, especially in relation to the measurement of cortisol levels. Cortisol concentration has only been obtained through the collection of blood, urine, or saliva, and such charges are of high financial cost.36
Gerber et al32 consider that capillary cortisol has the potential to fill the long-term methodological vacuum of cortisol assessment. In addition, capillary cortisol analysis may aid to a better understanding of how long-term cortisol elevation mediates stress-related effects on health and performance of recreational practitioners and athletes.
Despite the scarce data and some inconsistencies, the researchers found that cortisol concentrations in hair are associated with stress-related symptoms and psychiatric disorders (e.g. posttraumatic stress disorder), medical conditions that indicate chronic activation of the HPA and other life situations associated with a high risk of chronic stress. It is suggested that PA, adiposity and substance abuse may correlate with cortisol concentrations in hair. In contrast to measures of a short-term release of cortisol (saliva, blood and urine), smoking and oral contraceptive use appear to be associated with cortisol concentrations in the hair. It is indicated that in pregnant women, an increase of cortisol concentrations in hair in successive quarters is found by Wosu.33
Although adaptable in the short term, repetitive and chronic HPA axis stimulation and cortisol release may lead to allostatic loading. It is indicated that the hypothalamus secretes a corticotropin-releasing hormone (CRH). As a result, CRH activates the anterior pituitary gland by stimulating adrenocorticotropic hormone (ACTH). In sequence, due to stimulation of the adrenal cortex releasing cortisol.17,37
The advantage in relation to the use of the hair as a type of procedure to evaluate the cortisol concentration is the permission of a retrospective review of the HPA axis. This measurement is reliable in up to 6 cm of wire length, equivalent to six months of analysis. Another favorable point is the ease of storage and collection of the material, which can be stored in an envelope, kept in the environment without risk of degradation. Hair color does not interfere with cortisol levels, but there are disadvantages such as a decrease in cortisol concentration levels after 4 cm, 5 cm yarn growth due to the leaching phenomenon. Among studies there are controversies related to the quantity of material collected and still, there is not a defined protocol of the levels and values to be collected.15,24
Gerber et al32 have pointed out that is crucial that scientists consider whether their research questions can be adequately addressed, since regular vigorous exercise results in substantially increased levels of cortisol in hair.
CONCLUSION
In conclusion, putting together all the considerations of this review, it possible to conclude that the hair can be used successfully, as a specimen, to determine the concentration of the cortisol. This would have an importance to verify the response of the HPA axis to the PA.
CONFLICT OF INTERESTS
The authors declare that there is no conflict of interests regarding the publication of this paper.
ACKNOWLEDGMENTS
The authors gratefully acknowledge the support of the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) (financecode 001), the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), the Universidade do Estado do Rio de Janeiro (UERJ), and the Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ).








