INTRODUCTION
Browning is an essential process that can significantly impact the sensory and nutritional properties of various food products.1 It is a complex reaction that involves the gradual color change of food, which can be positive or negative, depending on the type of reaction. The two main types of browning reactions are enzymatic browning and non-enzymatic browning. Non-enzymatic browning occurs through chemical reactions that produce browncolored substances without the involvement of enzymes. This type of browning includes the Maillard and Caramelization reactions, which occur in various food products. However, non-enzymatic browning reactions usually occur in conjunction with other reactions, as food products contain complex constituents.2
On the other hand, enzymatic browning is caused by the polyphenol oxidase enzyme, which triggers the oxidation reaction in food products. This type of reaction is responsible for the texture, taste, flavor, aroma, and nutritional value of food. Enzymatic browning mostly occurs during harvesting, transportation, storage, and processing.3 The polyphenol oxidase enzyme involves two main reactions: the hydroxylation of mono-phenols to di-phenols, which is relatively slow and produces colorless products, and the oxidation of di-phenols to quinones, which is relatively fast and produces colored products. The oxidation reaction occurs when food products are exposed to air, which may happen due to physical or mechanical damage, such as cutting, peeling, slicing, and severe temperature changes during food processing and storage.4
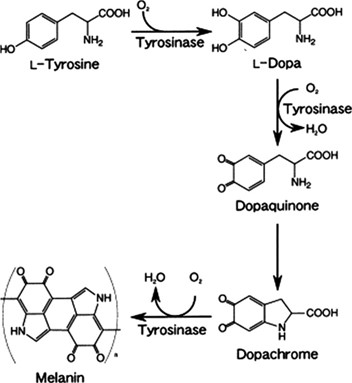
The browning reaction starts when the substrates and enzymes present in an apple are mixed in the presence of oxygen.5 When apple tissues are exposed to air, the oxidation of polyphenolic compounds is initiated; as a result, melanin (reddish-brown color compound) is gradually formed. The copper-containing enzymes polyphenol oxidase (PPO), also called mono-phenols, tyrosinase, and mono-oxygenase, act as substrates for the initiation of melanin formation depending on the substrate specificity.6 The metabolic pathway for melanin formation is initiated by tyrosine catalyzed by the polyphenol oxidase enzyme, as shown in Figure 1.
Figure 1. The Metabolic Pathway for the Formation of Melanin from L-Tyrosine Initiated by Polyphenol Oxidase
There are several physical and chemical methods or approaches for the preservation of apples from browning reactions, but in the present research, we mainly focused on the chemical treatments or methods for the inhibition of polyphenol oxidase enzyme activity by using different chelating agents, antioxidants, or reducing agents.
Pre-treatment to Apples
As we know, fruits and vegetables contain cuticle as a natural barrier for external attacks. Similarly, apples also contain cuticles composed of hydrophobic biopolymers with waxes, which act as a main limitation or barrier for the molecules to be diffused, whether used in chemical treatment or in physical methods. Therefore, we have to apply some pre-treatments to apples, such as different permeabilization methods involving the degradation of cuticle by using a solution of 2-8% ethyl oleate with 1-8% NaO.7 Also, permeabilization can be achieved by vacuum impregnation.8
Chemical Treatments – Chelating Agents as a Chemical
Treatment
Polyphenol oxidase PPO is a copper-containing enzyme belonging to the family of oxidoreductases; it requires the activation of copper ions for its normal enzymatic activity.9 Thus, we can inhibit the activity of PPO by adding some substances that have the capability of binding to divalent cations; as a result, copper ions cannot be activated, and ultimately, the PPO activity is inhibited. Kojic acid, EDTA E385, and citric acid E330 are the most common chelators used as chelating agents for the inhibition of PPO activities.
Antioxidants as a Chemical Treatment
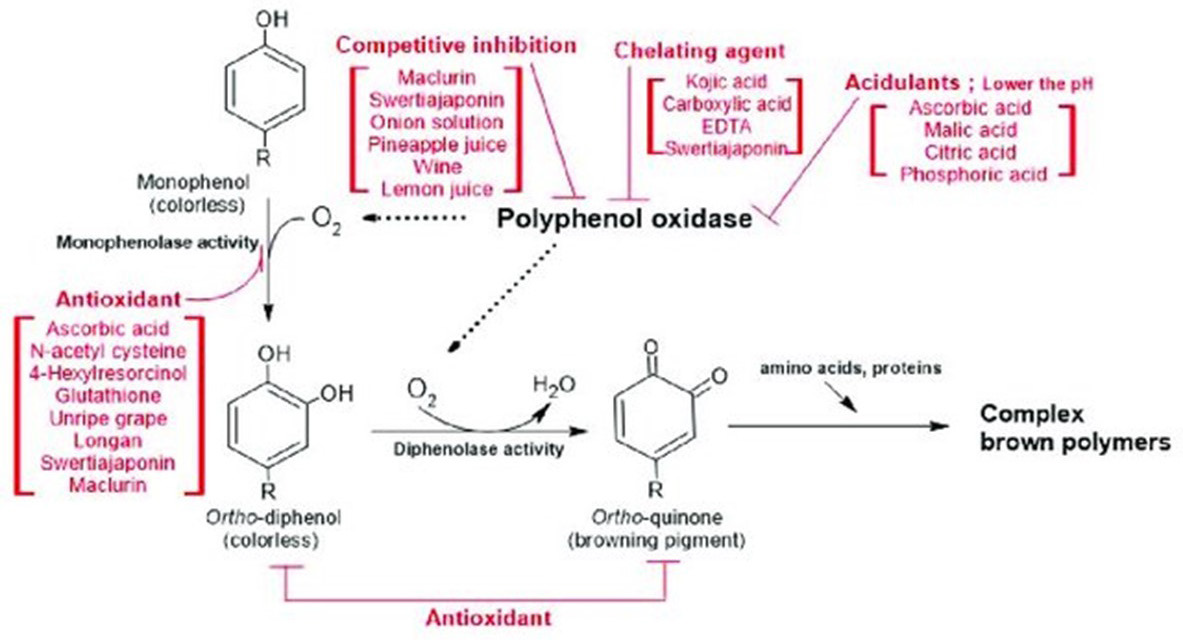
The formation of melanin (a reddish-brown substance) can be prevented by using different antioxidants, as antioxidants break the chain reaction by reacting with intermediate products of apples.10 Ascorbic acid, erthyorbic acid, glutathione, N-acetyl cysteine, etc. are the main antioxidants involved in the inhibition of PPO’s browning activity.11,12 The effectiveness of these antioxidants depends on various environmental factors, like pH, temperature, etc (Figure 2).
Figure 2. The Simplified Processes of Enzymatic Browning and the Inhibition Mechanisms of Antibrowning Agents
Agents of Firmness as a Chemical Treatment
We can use different agents of firmness to prevent the browning of apples, as these agents prevent the polyphenol oxidase from coming into contact with the polyphenol in the vacuole.13,14,15 For this purpose, we used calcium salts such as calcium lactate E327 and calcium chloride E509 for the firmness and strengthening of the cell wall.
Acidifying Agents as a Chemical Treatment
The polyphenol oxidase enzyme is naturally very sensitive to pH variations. Adding acids to apples may cause a reduction in their PPO activity or make them inactive if the pH level is below 3.16,17
For the prevention of apple browning, we used different chemical treatments, as shown in Tables, which either include antioxidant agents, acidifying agents, agents of firmness, or a combination of these agents. The oxidation reaction of PPO also depends on storage conditions because they might contribute to the efficiency of chemical agents and their chemical concentration solutions. Moreover, the higher efficacy of chemical preservatives is achieved when we use combinations of chemical treatments.
MATERIALS AND METHODS
Materials like fresh apple slices, beakers, mortar and pestle, paper plates, tongs, a timer, and a weighing machine were used in the experiment. Different types of reagents were used to determine the inhibitory activity of PPO. These are dilute water, potassium metabisulfite (KMS), sodium benzoate, and ascorbic acid. To begin the experiment, take the appropriate sample, use the weighing equipment to calculate the concentration, and prepare the solution before labeling the sample. In the next step, slice the apple into thin pieces and dip the apple slices into the solutions with tongs until completely saturated. Afterwards, remove the apple slices from the pan and set them on the paper plate. The concentration values must be noted on the paper plate. (To fight contamination, make sure to wash the tongs after every use.) The last step is to evaluate the browning effect. Keep the observations for 40 to 45-minutes.
RESULTS
Ascorbic Acid
Ascorbic acid was used to suppress the activity of polyphenol oxidases in sliced apples. The apple slices were cultured at various pH levels, temperatures, and intervals. At 0.35% concentration, ascorbic acid, better known as vitamin C, demonstrated significant inhibition of PPO. The browning of the apple was considerably affected at room temperature after 30-minutes. The high antioxidant activity of ascorbic acid, along with its capacity to create other elements and vital nutritional components, prompted food manufacturers to develop a specialized addition for usage in various foods.18 Phytochemicals apart from ascorbic acid (vitamin C) play an integral role in the anti-oxidant action of apples, along with the ability to suppress tumor cell growth. Room temperature was chosen as the optimal temperature for inactivating the enzyme. Ascorbic acid reacts with free radicals, inhibiting chain reactions that might have negative repercussions for organisms. At a level of 0.05%, no restriction of polyphenol oxidase was reported. After 15-minutes at room temperature, the apple adapted a golden brown color. The apple slices were treated with a 0.15% concentration of ascorbic acid at room temperature. PPO was found to be somewhat inhibited. In 20-minutes, the browning of the apple was somewhat minimized. The following testing was conducted at room temperature with a concentration of 0.20%, as shown in Table 1. PPO is inhibited to a modest extent. The browning of apples caused by enzymes was decreased to some degree.
| Table 1. Effects of Inhibition of PPO in Apple Using Different Concentrations of Ascorbic Acids |
|
Chemical Agents
|
Concentrations |
Temp/ Time |
pH |
Results |
Interpretation
|
|
Apple
|
Ascorbic acid
|
0.05%
|
RT /15 mins
|
2.60
|
No inhibition of polyphenoloxidase |
Apple turns into yellow brown quickly |
|
0.1%
|
RT /15 mins
|
2.56
|
No inhibition of PPO |
Apple turns into yellow brown quickly |
|
0.15%
|
RT/ 20 mins
|
2.52
|
Slight inhibition of PPO |
Browning of apple is slightly reduced |
|
0.20%
|
RT/ 20 mins
|
2.50
|
Inhibition of PPO is observed to some extent |
Moderate enzymatic browning of apple is observed |
|
0.25%
|
RT/ 25 mins
|
2.49
|
Sufficient inhibition of PPO |
Slow browning of apple noted |
|
0.3%
|
RT/ 25 mins
|
2.37
|
Significant inhibition of PPO |
Browning of apple is delayed |
|
0.35%
|
RT/ 30 mins
|
2.30
|
Prominent inhibition of PPO |
Browning of apple is reduced to some great extent |
| *RT=Room Temperature (30±5) |
Potassium Meta-Bisulfite
Another trial was made with a white crystalline powder, potassium metabisulphite. The major benefit of KMS is that it has antioxidant properties as well as the capacity to inhibit polyphenol oxidase, which catalyzes food browning. It’s used to keep fruits fresh. At 0.30%, the KMS showed visible inhibition of PPO. PPO inhibition was initiated with a 0.05% KMS concentration. During the experiment, the temperature was kept at room temperature. After 20-minutes, the moderate browning effects of the apple were noticed. PPO was not prevented at the lowest concentration, which was 0.002%, as shown in Table 2. Within 15-minutes, the apple had turned a golden-brown color.
| Table 2. Effects of Inhibition of PPO in Apple Using Different Concentration of KMS |
|
Chemical Agents
|
Concentrations |
Temp/ Time |
pH |
Results |
Interpretation
|
|
Apple
|
KMS or Potassium Meta-bisulfite
|
0.002%
|
RT /15 mins |
7.14
|
No inhibition of polyphenoloxidase |
Apple turns into yellow brown quickly |
|
0.003%
|
RT /15 mins |
7.21
|
No inhibition of PPO |
Apple turns into yellow brown quickly |
|
0.004%
|
RT/ 20 mins |
7.32
|
Slight inhibition of PPO |
Browning of apple is slightly reduced |
|
0.05%
|
RT/ 20 mins |
6.90
|
Inhibition of PPO is observed to some extent |
Moderate enzymatic browning of apple is observed |
|
0.1%
|
RT/ 25 mins |
5.51
|
Sufficient inhibition of PPO |
Slow browning of apple noted |
|
0.25%
|
RT/ 25 mins |
4.80
|
Significant inhibition of PPO |
Browning of apple is delayed |
|
0.30%
|
RT/ 30 mins |
4.50
|
Prominent inhibition of PPO |
Browning of apple is reduced to some great extent |
Sodium Benzoate
Another study was carried out using sodium benzoate at a concentration of 0.005%, as shown in Table 3. This is a very common food preservative. The Food and Drug Administration of the United States declared it GRAS. The maximum amount of this chemical that can be used is 0.1%. It has a little inhibitory effect on PPO. The browning of the apple has been mitigated to some extent at this point. There was no inhibition of PPO at 0.003% concentration, and at 0.002%, the apple became yellowbrown at a rapid rate.
| Table 3. Effects of Inhibition of PPO in Apple Using Different Concentration of Sodium Benzoate |
|
Chemical Agents
|
Concentrations |
Temp/ Time |
pH |
Results |
Interpretation
|
|
Apple
|
Sodium Benzoate
|
0.002%
|
RT/15 mins |
8.01
|
Inhibition of PPO is observed to some extent |
Moderate enzymatic browning of apple is observed |
|
0.003%
|
RT/ 20 mins |
8.13
|
Significant inhibition of PPO |
Browning of apple is delayed |
|
0.005%
|
RT/ 30 mins |
8.25
|
Prominent inhibition of PPO |
Browning of apple is reduced to some great extent |
Combination
In most cases, food preservation is dependent on the combined or synergistic actions of multiple additives. This mechanism prevents undesired product changes, reduces the need for preservatives, and restricts the number of procedures. Temperature, pH, and timing are examples of extrinsic parameters. The premise of blending additives allows for an infinite number of preservation possibilities. This is accomplished by acquiring foods that are nutritious and safe. A combination of both chemical agents, like potassium metabisulfite and sodium benzoate, was used to prevent the enzymatic browning. The combination demonstrated little suppression of PPO at 0.002%, as shown in Table 4. The presence of golden brown had only minor impacts. The combination inhibited PPO at a concentration of 0.006%. For the next 20-minutes, the golden-brown activity was entirely stopped.
| Table 4. Effects of Inhibition of PPO in Apple Using a Combination of KMS+Sodium Benzoate |
|
Chemical Agents
|
Concentrations |
Temp/ Time |
pH |
Results |
Interpretation
|
|
Apple
|
KMS+Sodium Benzoate
|
0.003%
|
RT/60 mins |
7.68
|
Inhibition of PPO is observed to some extent |
Slight enzymatic browning of apple is observed |
|
0.004%
|
RT/50 mins |
7.7
|
Minor inhibition of PPO |
Moderate enzymatic browning is observed |
|
0.006%
|
RT/ 100 mins |
7.9
|
Prominent inhibition of PPO |
Browning of apple is reduced to some great extent |
DISCUSSION
Liquids possess the characteristic of being either acidic, basic, or neutral in nature. The skin can be burned by strong acids and bases. During the experiment, appropriate observations were made regarding the use of chemicals. The pH scale was utilized to determine the acidity and basicity of these substances. The data obtained from laboratory-scale studies was analyzed, and previous research was taken into consideration. KMS has been used as an antioxidant and food preservative for centuries. With advancements in technology, food can now be preserved for extended periods without experiencing enzymatic browning. This preservation method is an initial step towards extending the shelf life of food. The human body was found to be adversely affected by higher dosages, whereas lower-levels were found to be beneficial. To apply this technique, a low-concentration solution must be sprinkled or sprayed on the cut apples, and it can last for up to two days.
CONCLUSION
The occurrence of enzymatic browning in apples has the potential to contribute to food scarcity, making effectiveness and profitability crucial factors. There are several food chemicals available that can serve as enhancers and possess desirable morphological characteristics. The most effective outcome was achieved by combining potassium metabisulphite and sodium benzoate at a concentration of 0.006%. The resulting mixture provided a coating for apple slices that effectively prevented browning while preserving their natural color. Sodium benzoate was also found to enhance the texture and flavor of the apple. This approach is considered safe for human consumption and has been classified as a generally regarded as safe compound (GRAS). It has a positive impact on apple preservation and can help to address global food shortages by extending the shelf life of apples.
ACKNOWLEDGEMENTS
This research work was supported by PCSIR Food and Biotechnology Research Center Lahore, so the authors are thankful to PCSIR Lahore for providing funds and for allowing them to conduct this research study.
INSTITUTIONAL REVIEW BOARD STATEMENT
This study was approved by the institutional review board (IRB) of PCSIR and the protocols used in this study were approved by the committee.
CONFLICTS OF INTEREST
The authors declare that they have no conflicts of interest.







