INTRODUCTION
BCG (Mycobacterium bovis BCG) is a vaccine for preventing childhood tuberculosis (TB). The World Health Organization registered freeze-dried Tokyo-172 seed lot as an international reference vaccine strain in 1965, and ever since, the Tokyo-172 BCG has been used, not only in Japan, but also all over the world. However, there are some severe adverse reactions that developed beyond vaccination site and regional ipsilateral axillary lymph-nodes. Osteomyelitis is a very rare but serious late complication of BCG-immunization and results from generalized dissemination of BCG.1 We present a case of a 23-month-old boy with a mass lesion in the right shoulder.
CASE REPORT
A 23-month-old Japanese boy with a mass lesion in the right shoulder was referred to our hospital. His parents noticed it and took him to hospital to consult. He was born by a normal vaginal delivery with a normal Apgar score, and was 82 cm tall, and weighs 9.8 kg now. He had no history of any diseases. On physical examination, the mass lesion was soft and slightly moved by touch. He could freely move his shoulder without pain or discomfort by it. His blood test and laboratory data showed elevated WBC (9600/μL (normal 3500-9000/μL) and LDH (318 IU/L (normal 106-211 IU/L), and showed decreased IgG 476 mg/dL (normal 800-1600 mg/dL), and IgA (44 mg/dL (normal 100-350mg/dL). CRP, IgM and others were normal. From these results, a chronic infectious disease due to weakened immune systems was suspected. On X-ray, questionable rounded radiolucent area with clear osteosclerotic margin was visualized in the proximal metaphysis to diaphysis of the right humerus (Figure 1). The right shoulder CT showed low density mass lesion in the subcutaneous fat tissue (Figure 2). The right humerus showed rounded radiolucent lesion with osteosclerotic margin (Figure 3). In order to check both the bone lesion and the subcutaneous soft tissue mass, we took right shoulder MRI. The subcutaneous mass lesion showed slightly high signal intensity on T2 weighted axial image (Figure 4a, arrow) and low signal intensity on T1 weighted axial image (Figure 4b, arrow). On contrast study, it showed ring enhancement (Figure 4c, arrow) suggestive of abscess formation. The proximal humerus showed low signal intensity on T1 weighted image (Figure 4b, arrowhead) and well enhancement on gadolinium-enhanced fat suppression T1 weighted axial image (Figure 4c, arrowhead). In addition, that enhanced area was connected with the proximal humeral lesion on gadolinium-enhanced fat suppression T1 weighted sagittal and coronal images (Figure 5a and 5b arrows). These findings suggested that both were the same lesion. We suspected chronic osteomyelitis with abscess formation of the right humerus from MRI finding and his symptoms. However, technetium-99 m MDP (250.0 MBq) bone scintigraphy showed no increased radioactive tracer in the right shoulder (Figure 6), suggestive of a cold abscess formation like tuberculosis. We asked his mother when and where on his body he was injected with the BCG vaccine. He had the Tokyo-172 BCG vaccine administered to his right shoulder when he was 2 weeks old. The abscess formation was present in the injection site of the vaccination of BCG. We diagnosed BCG-associated osteomyelitis and performed the subcutaneous abscess drainage and the curettage of the proximal humerus. Tuberculosis-polymerase chain reaction (Tb-PCR) was also performed from the subcutaneous abscess and it was positive. He received isoniazid (INH) and rifampins (RFP) therapy for one year. His right humerus was fully recovered. His immune systems became normal.
Figure 1. X ray of right humerus.
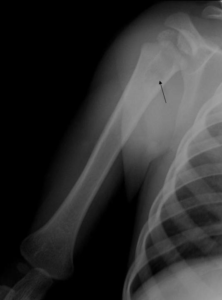
A rounded radiolucent area with clear osteosclerotic margin is visualized in the proximal metaphysis to
diaphysis of the right humerus (arrow). We cannot detect soft tissue mass in this radiograph.
Figure 2. Right shoulder CT (soft tissue density window) Low density mass lesion is
visualized in the subcutaneous fat tissue of the right shoulder joint (arrow).
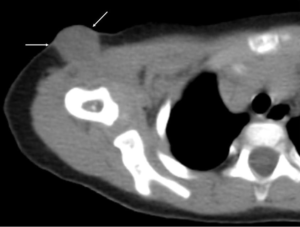
Figure 3. Right shoulder CT (bone density window) Low density area with osteosclerotic margin is present within the right humerus (arrow)
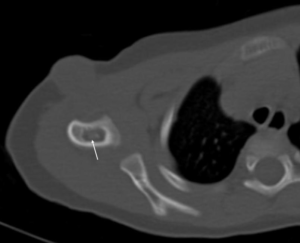
Figure 4. Right shoulder MRI: a. T2 weighted axial image, b. T1 weighted axial image, c. gadolinium-enhanced fat suppression T1 weighted axial image.
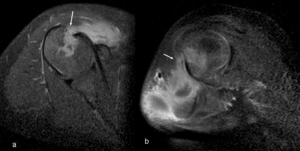
Subcutaneous soft tissue mass shows slightly high signal intensity on T2WI (a, arrow), and low signal intensity on T1WI (b, arrow). The bone lesion shows low signal intensity on T1WI (b, arrowhead). On contrast study, the subcutaneous soft tissue mass is ring-enhanced, suggestive of abscess formation (c, arrow). The bone lesion also shows enhancement (c, arrowhead).
Figure 5. Right shoulder MRI: Gadolinium-enhanced fat suppression T1 weighted axial image.

A saggital image, b. coronal image. Both the soft tissue mass and the bone lesion are connected
with each other (a, b arrows). Both are compatible with osteomyelitis with subcutaneous abscess formation.
Figure 6. Technetium-99 m MDP (250.0 MBq) bone scintigraphy.
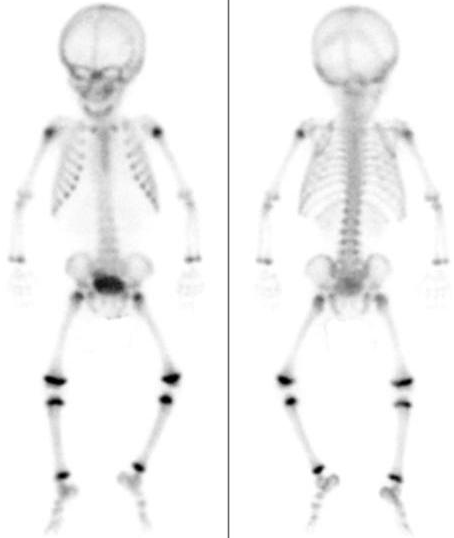
Whole bone scintigraphy shows no abnormal increased radioactive tracer.
The right shoulder lesion is suspious for cold abscess formation due to tuberculosis osteomylitis.
DISCUSSION
Outbreak of the BCG-associated osteomyelitis is influenced by a Japanese vaccination. In Japan, children were vaccinated with BCG until they reached the age of 4. Tuberculosis Control Law, which provides a legal basis for national tuberculosis control, was amended in 2004 and entered into force on April 1, 2005.2 After that, infants could have a BCG vaccination within 6 months after birth for 2005 to March 2013 to prevent tuberculosis. Therefore, a lot of infants had it at the same time when they received a medical check at 3 to 4 months after birth. Ever since, the rate of patients with BCG-associated osteomyelitis and ostitis increased by approximately 4 times in Japan (1.25 patients/year (2001-2004) vs. 4.14 patients/year (2005-2011).3 In addition, patients who were vaccinated the BCG within 4 months after birth were prone to be infected with the BCG-associated osteomyelitis and ostitis.
This might be because the immune system, especially cell-mediated immunity of the infants at 3 to 4 months after birth is immature yet.
Today, the amendment of the Preventive Vaccination has changed since April, 2013, and children must receive the BCG vaccine between ages of 5 months and 1 year. We have to know whether patients with the BCG-associated osteomyelitis will decrease or not.
Several authors reported that some children have primary immunodeficiency diseases and there are a number of primary immune deficiency diseases prone to BCG complications.4,5 Hoshina et al5 reported that patients who had multiple osteomyelitis diagnosed with interferon-γ receptor 1 deficiency from their genetic analysis. Development of osteomyelitis of patients with no gene abnormality was 10 months (1-46 months). It was late as compared to the patients with gene abnormalities (9.5 months) (7-15 months).6
In our case, the patient had the BCG vaccination at age of 5 months in 2012 and found the BCG-associated osteomyelitis as a non-painful mass 18 months later. From his laboratory data, he might be a temporary immunodeficiency state because he was too young when he was vaccinated. However, he didn’t have any primary immunodeficiency diseases or gene abnormalities. He has been growing up without any problems after his treatment.
The BCG-associated osteomyelitis occurs in long bones such as femur, humerus, and tibia. It does not necessarily occur in a bone which is close to the injected site of the BCG vaccination. X-rays of the BCG osteomyelitis show a lucent lesion with modelling deformity and a periosteal reaction. This fills in slowly with time and ongoing treatment. MRI is useful to detect BCG osteomyelitis with abscess formation. Bone scintigraphy is characteristic to the BCG osteomyelitis. We cannot detect any accumulation of radioactive tracer in the lesion. However, all image findings of the BCG osteomyelitis are extremely similar to ones of tuberculosis osteomyelitis.7 Therefore, we need to consider the possibility of tuberculosis osteomyelitis as well.
CONCLUSION
BCG-associated osteomyelitis is a rare complication after BCG vaccination. Infants who were vaccinated the BCG within 5 months after birth are prone to be infected with the BCG-associated osteomyelitis because their immune system is immature. We need to check their primary immunodeficiency diseases or gene abnormalities when we suspect the BCG-associated osteomyelitis.
CONFLICTS OF INTEREST
None.
CONSENT
Authors obtained written informed consent from the patient for publication of this manuscript.










