INTRODUCTION
Type 1 diabetes mellitus (T1DM) is a disease that compromises pancreatic function and requires strict glycemic control to minimize lifelong complications. These complications can have systemic manifestations that affect vision, renal function and have the potential to decrease the life span of those affected by 13 years.2 Education and constant vigilance are key factors in managing these complications. An organization called; Children with Diabetes, was created to educate affected individuals and their families about T1DM (http://www.childrenwithdiabetes. com). The participants take part in various sessions including a comprehensive non-mydriatic ocular screening program where attendees and their families learn about ocular health as it relates to diabetes. For the past decade, the Institute of Ophthalmology and Visual Science (IOVS) located at Rutgers State University (Newark, NJ, USA) has provided over 2,500 comprehensive eye exams remotely with a volunteer staff of medical professionals, medical students as well as optometry students (SUNY College of Optometry, New York, NY, USA) and pre-college students interested in the field of health care.
SUBJECTS AND METHODS
The comprehensive ocular screening was conducted following Health Insurance Portability and Accountability Act of 1996 (HIPPA) guidelines as well as an approved Institutional Review Board (IRB) at Rutgers University Hospital, Newark, New Jersey, USA. The screening was conducted in July 2016 and included 218 participants; 61% female and 39% male. The screening constituted of several stations divided between two rooms.
As seen in Figure 1, in the first room (Left) participants provide baseline information such as; body mass index (BMI), blood pressure (BP), visual acuity (VA), and pupil size measured with a pupilometer (Neuroptics, VIP™-200 Irvine, California, USA).
Figure 1: Flow Chart Depicting Participant Flow during Screening. Room on the Left: Participant General Information; Pupil Size, Visual Acuity, Blood Pressure, Pulse, and Participant Intake form Consisting of Height, Weight, BMI, Diabetes Management, Family and Past Medical and Ocular History. Room on the Right: Retinal Screening Room. Number Correspondence; 1- auto-refractor (Canon, RKF2 Tokyo, Japan), 2- tonometer (Canon, TX 20 Tokyo, Japan), 3- SD-OCT (Optovue, iVue, Fremont, California, USA),4-OCT-A (Canon, TX 20 Tokyo, Japan), and 5- (Canon, CR2 Plus-AF with EOS-60D Tokyo, Japan). Two Reading Stations with Trained Retinal Readers. Low Lighting was Present in Room #2 Allowing for Maximally Dilated Pupils without using a Mydriatic Agent.
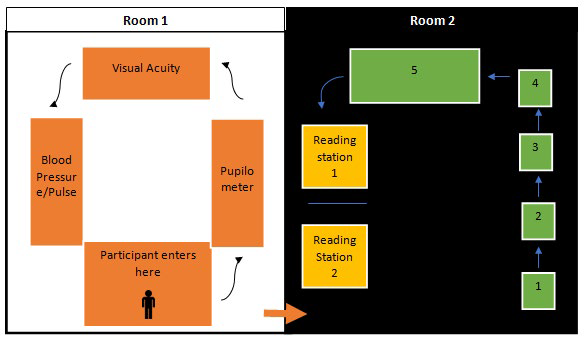
The second room (Figure 1, right side) was illuminated at a level of 80 candelas in order to maintain naturally dilated pupils for optimal retinal imaging. The room consisted of 6 stations: the first station was an auto-refractor (Canon, RKF2 Tokyo, Japan), the second station was an automated puff tonometer (Canon, TX 20 Tokyo, Japan), the third station was a Spectral Domain Ocular Coherence Tomography (SD-OCT) (Optovue, iVue, Fremont, California, USA), the fourth station was an Ocular Coherence Tomography-Angiography (OCT-A) (Optovue, AngioVue, Fremont, California, USA), the fifth station was a non-mydriatic retinal camera (Canon, CR2 Plus-AF equipped with a EOS-60D SLR camera with a 45 degree view and a resolution of 18.1 mega pixel APS-C CMOS, Tokyo, Japan). Retinal photography station was placed as the last testing station since it uses a Xeon flash tube that compromises natural pupil dilation (flash setting in color imaging was 80 Watt second while auto fluorescence was 300 Watt seconds). Station six was the reading/ evaluation station where all data was interpreted. This station had tele-ophthalmic connectivity through a DICOM connection to allow us access to an offsite board-certified ophthalmologist for instant consultation when needed.
All ocular testing was performed without use of mydriatic agents. Since our participants were predominantly children who were also partaking in outdoor programs we wanted to avoid dilation. Second, we wanted to avoid precipitating acute angle-closure glaucoma via the use of dilating agents. Lastly, dilating agents take about 20 minutes to be effective, our total screening is under 15 minutes per participant and this additional step would reduce the number of screenings we could provide.
Technology Advantages and Limitations in this Population
Auto-refractor/keratometer: Since this is a comprehensive ocular screening; automated refraction was used in addition to visual acuity (at 20 feet). Auto-refractors use retinal reflex alterations as the subjects fixate at a given target to determine at which point the image is at focus on the retina.3 Our screening used the Canon’s RK-F2 (Tokyo, Japan) which has several key features that allowed for ease of operation. First, once the subject’s head is positioned properly, the operator was able to fine tune eye alignment using an omni-directional joystick and capture bilaterally all the required data without any further operator intervention. This model had an additional advantage of including keratometry readings allowing for the simultaneous detection of astigmatism. Lastly, the 3D eye monitoring system in the ref-keratometer allows for maintenance of alignment irrespective of subject’s ocular movement allowing for ease of operation especially in a pediatric setting. The average autorefraction in our population were 0.25 OD and 0.50 OS. No age limitation was placed during this testing.
Automated puff tonometry: The automated non-contact puff tonometer used in this screening was a Canon, TX 20 (Tokyo, Japan) that measures the intraocular pressure (IOP) by delivering a measured puff of air into the subject’s cornea (apex) and determines the pressure of air required to momentarily flatten the apex of the cornea. Two important criteria for characterizing IOPs as abnormal are when the IOPs are higher than 21 mm Hg and when the difference between the right and left eye IOP exceeds 3 mmHg. The average IOPs in our population were 19.58 OD and 19.60 OS, both falling within the normal range of 10-21 mm of mercury. The importance of intraocular pressure in a diabetic population is highlighted in previous studies indicating a correlation between higher IOPs and elevated glycemic levels.4 We placed an arbitrary cut off age of 10-years-old strictly based on subjects cooperation.
Optical coherence tomography: The Ocular Coherence Tomography (OCT) (Optovue, Fremont, California, USA) has now become a standard component in all our retinal screenings. It’s advent and progress since its inception in 1991 have provided eye care professionals with the ability to visualize cross-sectional images of retinal layers and quantify the thickness of these layers with a resolution of 6 microns; retinal nerve fiber layer, ganglion cell complex, and macular area. Images are captured via the back reflectance of light waves and the time delay in this reflectance helps to characterize the spatial differentiation between the retinal layers. Incorporation of this particular instrument was critical to our screening due to its ability to detect early macular edema, a common consequence of diabetic retinopathy due to fluid leakage from weak retinal vasculature.5 No age limitation was applied to this testing.
Optical coherence tomography-Angiography: A new addition to our comprehensive retinal screening in T1DM started in 2015 has been the Optical Coherence Tomography Angiography (OCT-A). Traditional clinical modalities of vascular imaging systems have been fluorescein angiography (FA) and indocyanine green (ICG) imaging; however, their invasive approach associated with known risk factors and discomfort to subjects limit their use in remote telemedicine and pediatric setting such as our retinal screening.
Although, FA and ICG may provide effective methods in detecting neovascularization or vascular defects, they have limited capacity in providing 3D imaging and information specific regarding depth of a given pathology.6 The OCT-A utilized in our screening has a scanning speed of 70,000 A-scans per second providing depth of about 2.5 mm and allowing for imaging from the internal limiting membrane to the choroid. In the context of diabetic retinopathy, at our screening, three important observations helped in characterizing participants control of diabetes; maintenance of foveal avascular zone, density of perifoveal area, and presence of tortuosity in retinal vasculature. No age limitation was applied to this testing.
Non-mydriatic retinal camera: A Canon CR-2 non-mydriatic camera was used in our screening. The screening room (Figure 1 Right) was kept at 80 candelas to maximize natural dilation of pupils. Our pediatric population had a natural pupil size of over 6 mm; greater than the minimum pupil size required for artifact free images, 3.2 mm. In total, five posterior pole images were captured in each subject; optic nerve head (ONH) and macula centered right and left images and ONH centered auto fluorescence image in either the right or left eye. Interposition of these images (automated montage, Canon proprietary software) allowed for a 60-degree view of the posterior pole. All images were captured on a Canon, CR2 Plus-AF equipped with a EOS60D SLR (Tokyo, Japan) camera with a 45 degree view and a resolution of 18.1 mega pixel APS-C CMOS sensor. This sensor has a true red, green, and blue rendition giving us accurate posterior pole colors in the dark areas of the macula/fovea where about 90% of the light is absorbed whereas the optic nerve head reflects about 90% of the incident light therefore good detail is required to capture the fleshy appearance of this structure.
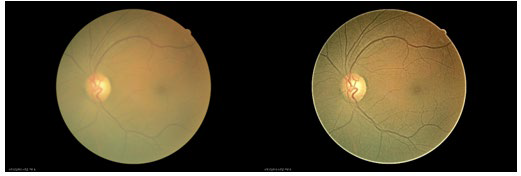
Reading and interpretation station : Post-screening, all data was made available either through electronic (intranet) or hard copy printout (vital signs). OCT, OCT-A, and retinal imaging was simultaneously displayed on color balanced computer monitor displays. Retinal images were evaluated for percentage of successful readable images. Success rate of imaging were scored based on the following criteria; clarity/sharpness, visualization of proper pre-selected fields, visibility of principal structures (macula, retinal vasculature, and optic nerve), and lack of external artifacts (blinking, lashes, poor centration of the camera, etc.). In situations where retinal images did not meet the image quality, a post-process “Opacity Suppression” digital filter (Canon, Tokyo, Japan) was to enhance images and enable the grader to read the images (Figure 2A, 2B). Retinal imaging success rate for the 2016 screening was 90%. Screened subjects and parents were offered copies of the screening results after all findings were thoroughly discussed with them.
Figure 2A, 2B: Left Side Image is of a 45° Field View of Retinal Image of Left Eye Taken in NonMydriatic Mode through a Mild Cataract. Right Side Images same 45° Field View of Retinal Image Post Capture Digital Processing Using Opacity Suppression (Canon, Tokyo, Japan).
RESULTS
In a 4-day period spanning over 30 hours and with 16 volunteer screeners, 218 subjects with T1DM were screened. Of these 218 subjects, 70 were repeat screenings for at least one year with 39 subjects that were at least 2 years consecutive follow-up. Only one subject that had been in previous years under control lost good glycemic and blood pressure control resulting in over 440 retinal hemorrhages in both eyes. After close follow-up of this subject over a period of 8 months post screening, this individual returned to normal retinal appearance. Participant demographics are shown in Table 1.
| Table 1: General Participant Demographics. |
|
Characteristic
|
|
Age (years) 19.13 (11-22)
|
19.13 (11-22) |
| Blood pressure (mmHg) |
111/70 (98/62-122/78)
|
|
Average pulse (beats/minute)
|
84 (73-94) |
| Average age of type 1 diabetes onset (years) |
8.7 (3.6-10.4)
|
|
Average duration of diabetes (years)
|
10 (4-13) |
| Average years of insulin pump use (years) |
7 (2.5-10)
|
|
Average years of CGM use (years)
|
3.45 (1.5-5) |
| Glucose (mg/dL) |
164 (112-200)
|
|
HbA1C (%)
|
7.57 (6.9-8.1) |
| Gender |
Male: 38.9%
Female: 61%
|
|
BMI
|
22.44 (18-25.7)
|
Our ocular screening included 218 participants, 61% female and 39% male. The ages ranged from 4 to 73 with an average of 19±12.8 years. The average bp was 111/70. HbA1C levels ranged from 5.1 to 13.0 with an average of 7.6±1.16. Average duration of diabetes for all subjects was 10 years. Subjects on average had been using insulin pumps and continuous glucose monitoring devices (CGM) for 7 and 3.5 years respectively. Of the 218 individuals screened, 10 had dot hemorrhages, 3 had flame hemorrhages and 1 had intraretinal microvascular abnormalities (IRMAs) affecting at least one eye. One subject required emergency referral to medical retina while 15 were referred for anterior segment evaluation for nuclear sclerosis.
DISCUSSION
Glycemic parameters have to be maintained within a limited range. Deviation beyond these parameters can result in profound adverse effects such as diabetic coma. Although, most screened individuals did well, one individual illustrates the importance of regular, consistent yearly screenings to better manage this chronic condition and prevent vision loss. The subject was a 21-year-old male who was diagnosed with T1DM in 1999. During his screening in 2012, blood glucose was 184 mg/dL, blood pressure was 128/78, and no indications of retinopathy were noted.7 He had adequate control for 13 years.
During his visit in 2013, his self-reported blood glucose was 421 mg/dL, self-reported HbA1c level was >13%, blood pressure was 130/91 mm Hg, and pulse was 106 bpm. Retinal findings included 2 small dot hemorrhages, no flame hemorrhages, and a single Intraretinal Microvascular Abnormality (IrMA). The subject reported inconsistent use of insulin intake and glucose control.
At the time of his screening visit in 2015 (15 years post diagnosis), he had been prescribed EnalaprilTM by his generalist for blood pressure control. However, he reported inconsistent use of Enalapril due to the development of numbness and pain in his legs which he attributed to the medication. He stopped taking Enalapril™. Since his previous visit, he had also altered his method of insulin administration from relying on a Medtronic™ insulin pump to self-administration of insulin subcutaneous injections due to the discomfort caused by the tubing in the Medtronic™ pump. His diet consisted primarily of fast food and carbonated sugar base drinks. These life style modifications resulted in a drastic change in both his physiological levels as well as retinal findings. His blood pressure at the time of his screening had increased to 142/62 mmHg, fasting blood glucose was 319 mg/dL, and HbA1c level was >13.5%. Retinal changes were severe with a total of 440 dot-blot and flame hemorrhages in the right and left eye primarily localized within the posterior pole consistent with diabetic retinopathy (Figure 3A and 3B).7 The OCT-A images showed signs of retinal telangiectasia and micro-aneurysms. The subject had developed non-proliferative diabetic retinopathy, but his OCT-A findings indicating an avascular foveal pit impacted the direction of management therapy to favor more behavioral changes rather than ocular clinical treatment to prevent disease progression (Figure 4).
Figure 3: (A) 45° Field View of Retinal Image of Left Eye Taken in Non-mydriatic Mode in July, 2015. (B) Center Image is a Montage Done without Dilation Comprised of 5 Images Captured in December 2015. (C) 45° Field View of Retinal Image Taken of Left Eye Taken in Non-mydriatic Mode in July, 2016.
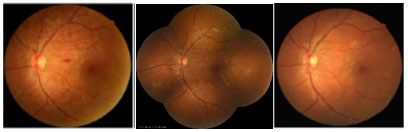
Figure 4: Top: OCT-A Image of Left Eye Taken July, 2015. Bottom: OCT-A Image of Left Eye Taken in July, 2016.
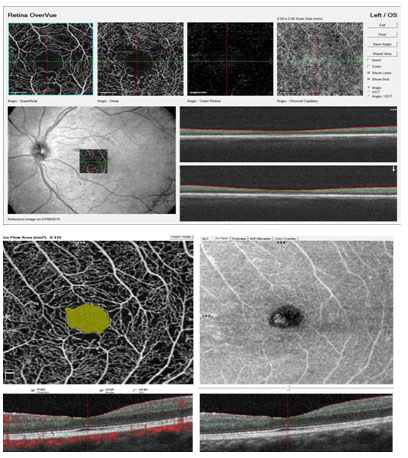
Figure 5: “Children with Diabetes” Screening Team 2016, July 2016. Front Row: C. Khouri, B. Szirth, K. Duong, M. Shah, L Deeb, L. Thangamathesvaran, Back row: S. Mendez, K. Soules, B. Peach, R. Ragam, K. Dunlap, J. Stroud, R. Freund, H. Cope, M. Ferm, C. Weng. Photo credit: Corporate Image Photography.

After the 2015 retinal screening, the subject revised his approach to his diabetes management and resumed taking Enalapril for blood pressure control, MedtronicTM insulin pump for glycemic control, and started to implement healthier life style modifications. Over the course of 8 months, the subject’s progress was monitored monthly both via retinal imaging (photography and OCT) and glycemic parameters. At the 2016 screening, his HbA1c had decreased to 8.1%, blood pressure was 110/75 mmHg, and he had two dot hemorrhages, no flame hemorrhages, and no IRMAs (Figure 3C). His positive progression emphasizes the importance of good follow-up with multi-specialty services as well as yearly retinal screenings that can monitor good glycemic and blood pressure control.
CONCLUSION
Fast paced ocular health screening programs (15 minutes) as it relates to T1DM can be successfully performed in subjects as young as 4 years of age. A non-mydriatic approach makes the experience more pleasant and convenient for the individual without compromising the quality of the images. Yearly comprehensive ocular health screenings along with T1DM related general education plays an important role in good vision control in T1DM while reminding individuals that screenings are not meant to be a replacement of a timely visit to the multi-specialty centers that can best control T1DM. One of the goals of Friends for Life yearly events is to create a safe learning family experience focused on establishing a lifelong support of all aspects of living with T1DM.
ACKNOWLEDGEMENT
This paper did not require an IRB; however, we did follow all guidelines from Health Insurance Portability and Accountability Act (HIPAA).
CONFLICTS OF INTEREST
None of the authors have a conflicts on interest.










