INTRODUCTION
Management of severe sepsis and septic shock has evolved over the past decade with optimization in the Surviving Sepsis Campaign’s (SSC) resuscitation bundle. The resuscitation bundle comprises a set of diagnostic and therapeutic approaches, which, when utilized, can act synergistically, leading to a greater survival benefit. The SSC started in 2002 to enhance clinician compliance and reduce mortality.1
The resuscitation bundle includes keeping maintain mean arterial pressure (MAP) ≥65 mm Hg, central venous oxygen saturation ≥70%, and central venous pressure ≥8 mm Hg. Following the diagnosis of severe sepsis versus septic shock, compliance was assessed at 3 hours and 6 hours.2 Several prior studies have concluded that there has been an inversely proportional relationship between compliance with severe sepsis/septic shock bundles and mortality.3 Several retrospective cohort studies nationwide have allowed for improvement in compliance with the utilization of the sepsis bundles, thereby reducing mortality and morbidity. Previous studies have indicated that adherence to the resuscitation bundles at 6 hour versus 18 hour completion was related to a significant reduction in mortality.4 Among several adults with a diagnosis of severe sepsis versus septic shock in the emergency department, hospital mortality decreased from 21.2 to 8.7% in 6 years. Simultaneous bundle compliance increased from 4.9-73.4%.2 In another study, it was observed that sepsis bundle implementation programs led to improved compliance with the 6 hour bundle (OR=4.12 (95% confidence interval 2.95-5.76) and 24 hour bundle (OR=2.57 (1.74-3.77) furthermore leading to a reduction in mortality (OR=0.66 (0.61-0.72).1 However, recent studies focusing on 3 hour versus 6 hour bundles proved beneficial for reducing mortality and cost-effective.5
This study was a retrospective cohort study that collected data on patients visiting the emergency department with a primary diagnosis of severe sepsis or septic shock. The study was conducted for 5 years from 2016 to 2021. The goal of this study was to analyze the mortality rate with the comparison of 3 hour versus 6 hour SSC bundles.
METHODS
Study Design
The study focused on adults 18 years and older with a diagnosis of severe sepsis or septic shock. Consistent with SSC guidelines, it assesses lactate measurement, obtaining blood cultures, administering broad-spectrum antibiotics, fluid resuscitation, vasopressor administration, reassessing volume status and tissue perfusion, and repeat lactate measurement. As reflected in the data elements and their definitions, the first three interventions should occur within 3 hours of the presentation of severe sepsis, while the remaining interventions are expected to occur within 6 hours of the presentation of septic shock. The rationale behind implementing the sepsis bundles was based on decreasing organ failure, overall reductions in-hospital mortality, length of stay, and care costs.
Data Collection
Retrospective data sources for required data elements included administrative data and medical record documents. This approach provided an opportunity for improvement at the point of care/ service. The data was collected using the assigned International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) codes. Coders were used to verify the accuracy of the diagnoses and codes. The sample was all patients who were in the emergency department with a primary diagnosis of sepsis versus septic shock. Inclusion criteria included: Discharges aged 18 and over with an ICD-10-CM principal or other diagnosis code of sepsis, severe sepsis, or septic shock. Excluded populations included coronavirus disease-2019 (COVID-19), comfort measures/ palliative care within 6 hours of presentation of severe sepsis and septic shock, administrative contraindication to care within six hours of presentation of severe sepsis and septic shock, length of stay >120-days, transfer in from another acute care facility, patients enrolled in a clinical trial for sepsis, severe sepsis or septic shock treatment or intervention, patients with severe sepsis or septic shock who are discharged within six hours of presentation, patients receiving 4 antibiotics for more than 24 hours before presentation of severe sepsis. Improvement was noted as an increase in the rate of compliance. A figure was calculated based on patients who received ALL of the following: Within 3 hours of presentation of severe sepsis: Initial lactate level measurement, broad spectrum or other antibiotics administered, blood cultures drawn before antibiotics, AND received within 6 hours of presentation of severe sepsis. ONLY if the initial lactate is elevated: Repeat lactate level measurement AND within 3 hours of initial hypotension: Resuscitation with 30 mL/kg crystalloid fluids OR within three hours of septic shock.
Statistical Analysis
Considering we have data that doesn’t conform to the traditional normal distribution, the statistical significance was confirmed using the Mann-Whitney U-test as appropriate. The resulting pvalues less than 0.05 were considered statistically significant. All statistical analysis was performed using Microsoft Excel software.
RESULTS
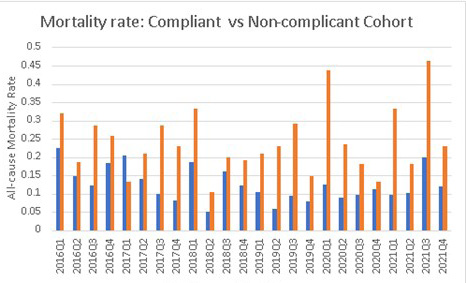
The final study cohort consisted of 2356 patients who met the inclusion criteria, and close to 76% of the study cohort confirmed compliance with the sepsis bundle. The computed mortality rates for the compliant and non-compliant cohorts were 12.3% and 23.6% respectively (p=1.8×10-6) (Figure 1 and Table 1).
Figure 1. Quarterly Mortality Rate Comparison from 2016 to 2021
| Table 1. Yearly Total Count and All-Cause Mortality Rates |
|
Year
|
Mortality |
Comp |
Mortality NC |
Non-Comp |
Total |
| 2016 |
33 |
192 |
27 |
101 |
293
|
|
2017
|
35 |
269 |
24 |
118 |
387 |
| 2018 |
27 |
203 |
24 |
108 |
311
|
|
2019
|
30 |
352 |
23 |
108 |
460 |
| 2020 |
35 |
331 |
15 |
59 |
390
|
|
2021
|
59 |
438 |
22 |
77 |
515 |
| Sum |
219 |
1785 |
135 |
571 |
2356
|
|
Comp
|
Non-Comp |
p value |
| Rate |
0.122689076 |
0.23642732 |
1.8×E-06
|
| Comp: compliant; NC: Non-compliant |
DISCUSSION
Our study showed a remarkable reduction in all-cause mortality rate from sepsis bundle compliance. Limitations to the study include varying endpoints, sources of sepsis, comorbidities, demographics, and a very high compliance rate. The differing endpoints may have skewed or further complicated our results because following patients after discharge is limited due to difficulties accessing medical records outside of our institution. The differing sources of sepsis can also complicate mortality rates since their fatality rates aren’t usually equal. The demographics and comorbidities would somewhat limit the generalizability of the study; however, our results are very well-applicable to the patient population we serve at our institution. Additionally, the unequal proportion between cohorts may have skewed the results. However, from the sampling standpoint, selection bias is minimal because sampling was conducted consecutively given the participant meeting the inclusion criteria.
Factors that affect data collection vary. Examples would be medical record completion, outstanding queries, and abstractor errors, to name a few. We have a process to validate abstractor accuracy, including reviewing a sample of completed cases every quarter. We attend all CMS updates on the measure and have access to a Q&A tool for clarification.
In the future, it would be recommended to perform morbidity and mortality statistics every year, especially in keeping compliance with the sepsis bundle. Future studies could account for consistent sources of sepsis and comorbidities. Furthermore, it would also be recommended to follow-up with patients after discharge to analyze the long-term benefits of the sepsis bundle compliance on the morbidity and mortality rates.6
CONCLUSION
We demonstrated that sepsis bundle compliance is strongly associated with a reduction in all-cause mortality. Importantly, it will be useful to identify further the specific bundle component with the strongest mortality reduction benefit.
INSTITUTIONAL BOARD PERMISSION
This research was conducted adhering to the ethical guidelines outlined on the journal’s author guidelines page. As the study did not involve human or animal subjects, no formal ethical approval was necessary.
CONFLICTS OF INTEREST
The authors declare that they have no conflicts of interest.






