INTRODUCTION
The brainstem is defined as the midbrain, pons and medulla; brainstem gliomas are generally diffuse intrinsic tumors involving the pons, with defined clinical presentation and characteristic appearance in imaging findings and do not require pathological confirmation;1,2,3,4 it can extend along neural tracts to adjacent regions of the brain, so it has been defined as fatal disease. Radiation treatment response rates show low degrees of efficacy, with short-term responses and a median overall survival less than one year;2,3,4,5,6,7,8 Diffusely infiltrating pontine gliomas must be distinguished from other subsets of diffuse intrinsic pontinegliomas, such as focal tumors, which are described with better prognosis and longer term survival.5,9
The association of Chemotherapy (ChT) and radiotherapy (RT) have not improved survival1,6,7,8,10,11 and now biologics are combined with RT in clinical trials.9,12,13
We investigated the association of RT with Nimotuzumab, a humanized monoclonal antibody developed at the Center of Molecular Immunology, Havana, Cuba and testing the hypothesis that this combination will improve survival in these tumors. The antibody was obtained by humanization of the murine antibody EGF/R3.14 Because Nimotuzumab has a 10 fold lower affinity to the EGFR, as compared to cetuximab, its capacity to bind EGFR is heavily dictated by cell receptor density.15 Nimotuzumab preclinical and clinical characterizations have been summarized before.16,17,18
A distinguishing feature of Nimotuzumab compared to other mAbs of the EGFR class is the lack of severe skin toxicity and the possibility to be used beyond progression.16,19
MATERIAL AND METHODS
We conducted a prospective, non-randomized clinical study, with a treatment group of 28 children and adolescents (range 3 to 18 years) with the diagnosis of diffuse intrinsic pontine glioma (DIPG ) documented by imaging (MRI, CT scan.) Biopsy and histology confirmation were not a requirement of this study. This study covers a sequential period between Jan/2009 and Dec/2012. Follow-up continued to September 2014. Patients with focal lesion of the brainstem were not eligible for the study, because these are described with a better prognosis.5,9 Also excluded were those who had received prior chemotherapy or radiotherapy.
The study was approved by the Ethical Institution Committee at the National Oncology and Radiobiology Institute (INOR), Havana and informed consent obtained from the patients’ parents. All patients were irradiated at INOR, and received the monoclonal antibody Nimotuzumab at INOR or at the Pediatric Hospital “Juan Manuel Marques”, also in Havana. Male patients: 14, and female: 14.
No surgical treatment was feasible, and in 3 there were pathologic results by biopsy. In all patients tumor was extended to the pons.
Linear Accelerator was used for a radiation treatment: Gross Tumour Volume (GTV) was defined as the visible tumor, either by MRI or CT; Clinical Target Volume (CTV) accounted for subclinical microscopic disease and unappreciated tumor extension, generally 1.5 cm from the GTV, and Planning Target Volume (PTV) was 0.3-0.5 cm and could vary according to the Organ at Risk.4,20 RT doses range from 54 to 59.8 Gy, with dose per fraction 1,8 Gy. Three patients were planned with IMRT and the rest with 3D CT. In all patients a thermoplastic mask was fitted.
Nimotuzumab was administered at a dose of 150 mg/m2 (IV), weekly during the term of RT, then every 2 weeks for 8 doses, then monthly for one year. In the last patients included it was prolonged for 2 years.
Twenty four patients (85.7%) received the complete Nimotuzumab schema, in 4 there were minor interruptions of the dosage because patients did not concur with the treatment option in a timely manner. Characters of the series are in Table 1.
Table 1: Charts of the series of brainstem gliomas in children and adolescents.
Median age at diagnosis……………….. 7 years old ( range 3-18 years )
Male/female ratio………………………… 1:1
Type of tumor:……………………………DIPG:28
Localization……………………………………Mid brain and pons: 15
Pons:6
Cerebello-pontine: 4
Not specified: 3
Pathology…………………………………………Grade II Astrocytoma:2
Grade III Astrocytoma: 1
Not biopsied: 25
Radiotherapy dose received……………………………54-57 Gy : 4 patients
57-59
Gy: 15 patients
59, 8 Gy: 9 patients
Nimotuzumab treatment…………………………….Completed: 24
Not completed: 4
Univariate and multivariate Cox regression models were fitted in order to identify possible predictors of survival and Kaplan Meier survival was calculated for all cases.
RESULTS
Median age of the cases at diagnosis was 7 years (range between 3 and 18 years old). Twenty eight cases were DIGP, but in all cases tumor extended to pons, and in one case also to 4th ventricle, cerebellum and hypothalamus.
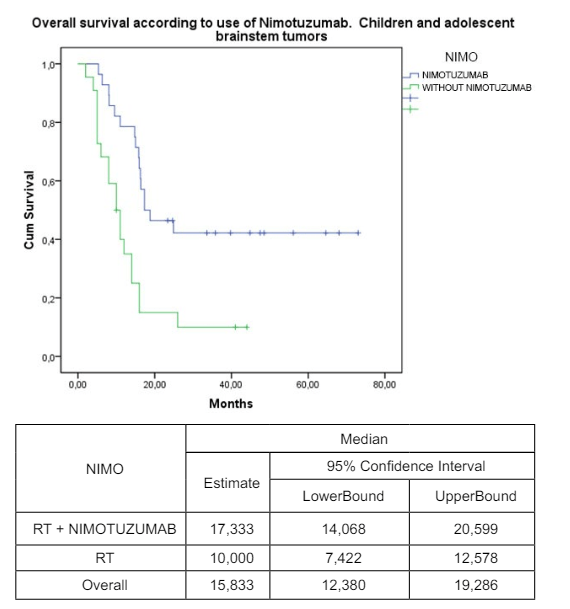
The diagnosis was made by clinical and imaging findings except in 3 cases where pathological examination was also done, one with astrocytoma grade II and the 2 others with astrocytoma grade III, all alive. Median overall survival from the beginning of treatment was 17,3 months, 95% CI (14.1-20.7) and Kaplan Meier survival accumulated was 42,9% at 2 years, established till 5 years of RT treatment. At present, (January 2015) 12 of the patients are alive (2 less than 2 years, 3 between 2-3 years, 5 between 4-5 years and 2 with more than 5 years of follow-up). Therapeutic results were compared with a previous case series of 22 children and adolescents with BSG treated with the same mode of irradiation, but without Nimotuzumab during 1992-2008 at the same institution (Instituto Nacional de Oncologia y Radiobiologia in Havana.) (Figure 1)
Figure 1: Survival according to treatment: a) 28 patients treated with Radiotherapy (RT) and Nimotuzumab, with a median overall survival of 17,3 months (95% CI 14.0-20.5) and accumulated survival of 42,9% at 5 years. b) Previous serie of 22 patients trated only with RT, with a median overall survival of 10 months (95% CI 7.4-12.5), with no survival accumulated at 5 years. P=0,001.
Sex
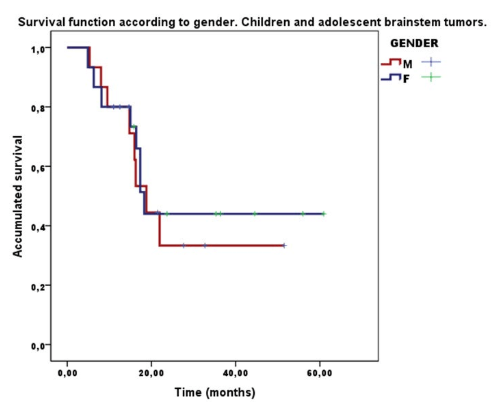
14 female patients had a 44.0% survival at 2 years and counting, and 14 male patients a 33.3 % survival. (Figure 2)
Figure 2: Survival function according to gender. 14 female patients, 44% survival at two years; 14 male patients patients, 33,3% survival at two years. No differences p=0.74.
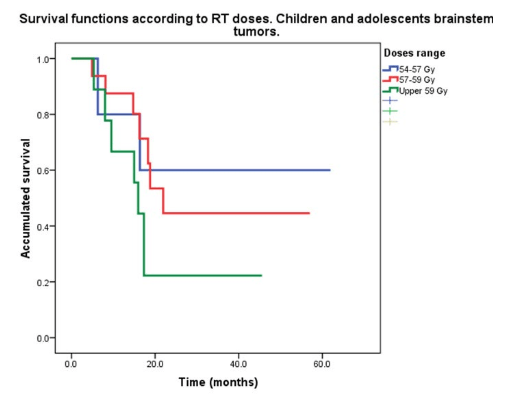
Five patients received a dose between 54-57 Gy, with a survival rate of 60% at 2 years and counting; 16 patients with a dose between 57-59 Gy with a survival rate of 41,2 % at 2 years plus, and 7 patients received 59.8 Gy with a survival rate of 22.2% at 2 years plus. Radiation dose was increased in those patients where DIPG tumor size exceeded the mean. (Figure 3)
Figure 3: Survival functions according to Radiotherapy (RT) doses: 5 patients received dose between 54-57 Gy with a survival rate of 60%; 16 patients with dose between 57-59 Gy with a survival rate of 41,2%, and 7 patients received 59,8 Gy with a survival of 22.2%; p=0254.
By Age Group
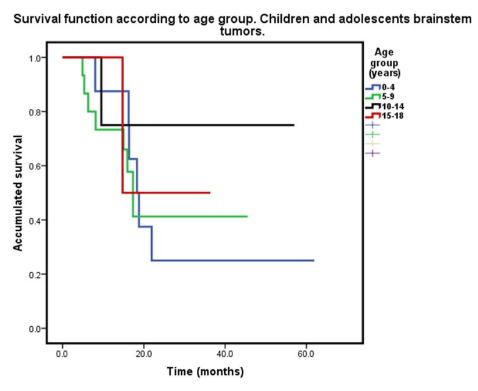
6 patients in the group up to 4 years old (21.5%) with a survival rate of 25% at 2 years plus; 15 patients between 5-9 years old (53.6%) with a survival rate of 37.5% at 2 years plus; 4 patients between 10-14 years old (14.4%) with a survival rate of 67%; and 3 patients with 15-18 years old age (10.7%) with a survival rate of 50% at 2 years plus (Figure 4).
Figure 4: Survivalo according to age group: 6 patients up to 4 years old with a survival rate of 25 %; 15 between 5-9 years old with a survival rate of 37,5%; 4 patients between 10-14 years old with a survival of 68% and 3 with 15-18 years old and a survival rate of 50%; p=0.777.
Multivariate analysis showed no significant differences according to sex, age and dose of irradiation received, nor with the dosage of Nimotuzumab, with a tendency for best survival in age group 10-18 years old and with the dose at 54-57 Gy.
Treatment was well tolerated and no grade III or higher grade toxicity was observed. The most frequent adverse event was alopecia in irradiation fields, observed in 27 patients (96.4%). Other reported adverse events were vomiting, headache, fever, tremor and nausea, in less than 40% of patients (Table 2).
| Table 2: Severity grade goes from 1 (less severe) to 4 (most severe). |
|
Most frequent related adverse events
|
|
Severity grade:
|
1 |
2 |
3 |
4 |
| Vomiting |
5 |
3 |
0 |
0
|
|
Headache
|
3 |
1 |
0 |
0 |
| Fever |
3 |
0 |
0 |
0
|
|
Tremor
|
0 |
2 |
0 |
0 |
| Nausea |
2 |
0 |
0 |
0
|
At the end of irradiation there was a clinical response in 27 patients (96.4 %), only one patient had no response to RT treatment.
DISCUSSION
Brainstem gliomas are a heterogeneous group of tumors that occur predominately in children, have a characteristic appearance in MRI and CT and do not require pathological confirmation: clinical and imaging are considered sufficient for diagnosis;1,2,3,4,21 they have a bad prognosis despite initial response to irradiation, which has been the ideal treatment, with a median survival of one year or less.1,2,3,4,5,12,21 Radiation dose escalation increases toxicity but does not improve outcome; neither chemotherapy nor hypofractionation4,21,22,23,24 have improved therapeutic ratio.
Brainstem gliomas could be diffuse intrinsic pontine gliomas (DIPG) and comprise an entity defined by imaging involving pons. Pontine localization was found to be a negative prognostic factor.5 Two more recent studies did not demonstrate similar findings;25,26 these studies included adult patients, possibly confounding results. In our series, we found pontine infiltration in all patients.
Our series of 28 children and adolescents with prolonged treatments involving Nimotuzumab in conjunction with RT showed a median overall survival of 17.3 months and a survival rate at 2 years of 39.7%, exceeding those of earlier reports,2,24,27 and of more recent ones.1,3,5,12,28,29,30,31,32,33,34 In a previous case series of patients treated only with RT during the period 1992-2008 in the same Institution (National Oncology and Radiobiology Institute, in Havana), median survival time was 10.0 months and the survival rate at 2 years was 15%. We know that the use of retrospective case series as control groups have limitations, but there are facts that are relevant: all cases were irradiated in the same Institution, so there could not exist center-effects that could bias patient groups.
It is remarkable that in our series there is a preliminary benefit to survival in older ages; it contrasts with reports that young age may predict a better outcome for children with diffuse pontinegliomas.34 Best results were obtained in the group that received 54-57 Gy of RT dosage; increasing RT dosage did not yield better results, consistent with.22,30
The prolonged use of Nimotuzumab for one year or more was well tolerated: adverse events such as alopecia and dermatitis were related to irradiation. Cutaneous rash was also reported but not related; other minor adverse events could be related in part with the clinical evolution of the tumor and the radiation treatment. Nimotuzumab is a humanized monoclonal antibody that recognizes the EGF external domain with intermediate affinity (kd=10-8 )35 that has shown a benefit in addition to RT in different studies.35,36,37 In children it has been reported with good results and safely associates to RT in CNS and Head and Neck tumors.2,31,32,33,34,35,36,37,38,39,40,41,42,43,44
CONCLUSION
The association of focal RT and Nimotuzumab in the treatment of brainstem tumors in children and adolescents was a therapeutic option in order to increase overall survival and support the possibility of new trials with this combination. Radiation dose over 57 Gy did not increase possibilities of survival in our series.
CONFLICTS OF INTEREST
No potential conflict of interest exists.









